| Colorectal cancer | |
|---|---|
| Other names |
|
 | |
| Location and appearance of two example colorectal tumors | |
| Specialty | Gastroenterology General surgery Oncology |
| Symptoms |
|
| Causes | Lifestyle factors and genetic disorders[2][3] |
| Risk factors |
|
| Diagnostic method | Tissue biopsy during a sigmoidoscopy or colonoscopy[1] |
| Prevention | Screening from age of 45 to 75 |
| Treatment | |
| Prognosis | Five-year survival rate 65% (US)[6] |
| Frequency | 9.4 million (2015)[7] |
| Deaths | 551,000 (2018)[8] |
Colorectal cancer (CRC), also known as bowel cancer, colon cancer, or rectal cancer, is the development of cancer from the colon or rectum (parts of the large intestine).[5] Signs and symptoms may include blood in the stool, a change in bowel movements, weight loss, abdominal pain and fatigue.[9] Most colorectal cancers are due to lifestyle factors and genetic disorders.[2][3] Risk factors include diet, obesity, smoking, and lack of physical activity.[2] Dietary factors that increase the risk include red meat, processed meat, and alcohol.[2][4] Another risk factor is inflammatory bowel disease, which includes Crohn's disease and ulcerative colitis.[2] Some of the inherited genetic disorders that can cause colorectal cancer include familial adenomatous polyposis and hereditary non-polyposis colon cancer; however, these represent less than 5% of cases.[2][3] It typically starts as a benign tumor, often in the form of a polyp, which over time becomes cancerous.[2]
Colorectal cancer may be diagnosed by obtaining a sample of the colon during a sigmoidoscopy or colonoscopy.[1] This is then followed by medical imaging to determine whether the disease has spread.[5] Screening is effective for preventing and decreasing deaths from colorectal cancer.[10] Screening, by one of a number of methods, is recommended starting from the age of 45 to 75. It was recommended starting at age 50 but it was changed to 45 due to increasing numbers of colon cancers.[10][11] During colonoscopy, small polyps may be removed if found.[2] If a large polyp or tumor is found, a biopsy may be performed to check if it is cancerous. Aspirin and other non-steroidal anti-inflammatory drugs decrease the risk of pain during polyp excision.[2][12] Their general use is not recommended for this purpose, however, due to side effects.[13]
Treatments used for colorectal cancer may include some combination of surgery, radiation therapy, chemotherapy, and targeted therapy.[5] Cancers that are confined within the wall of the colon may be curable with surgery, while cancer that has spread widely is usually not curable, with management being directed towards improving quality of life and symptoms.[5] The five-year survival rate in the United States was around 65% in 2014.[6] The individual likelihood of survival depends on how advanced the cancer is, whether or not all the cancer can be removed with surgery, and the person's overall health.[1] Globally, colorectal cancer is the third most common type of cancer, making up about 10% of all cases.[14] In 2018, there were 1.09 million new cases and 551,000 deaths from the disease.[8] It is more common in developed countries, where more than 65% of cases are found.[2] It is less common in women than men.[2]
Signs and symptoms
[edit]The signs and symptoms of colorectal cancer depend on the location of the tumor in the bowel, and whether it has spread elsewhere in the body (metastasis). The classic warning signs include: worsening constipation, blood in the stool, decrease in stool caliber (thickness), loss of appetite, loss of weight, and nausea or vomiting in someone over 50 years old.[15] Around 50% of people who have colorectal cancer do not report any symptoms.[16]
Rectal bleeding or anemia are high-risk symptoms in people over the age of 50.[17] Weight loss and changes in a person's bowel habit are typically only concerning if they are associated with rectal bleeding.[17][18]
Cause
[edit]75–95% of colorectal cancer cases occur in people with little or no genetic risk.[19][20] Risk factors include older age, male sex,[20] high intake of fat, sugar, alcohol, red meat, processed meats, obesity, smoking, and a lack of physical exercise.[19][21] The Rectal Cancer Survival Calculator developed by the MD Anderson Cancer Center additionally considers race to be a risk factor; however, there are equity issues concerning whether this might lead to inequity in clinical decision making.[22][23] Approximately 10% of cases are linked to insufficient activity.[24] The risk from alcohol appears to increase at greater than one drink per day.[25] Drinking five glasses of water a day is linked to a decrease in the risk of colorectal cancer and adenomatous polyps.[26] Streptococcus gallolyticus is associated with colorectal cancer.[27] Some strains of Streptococcus bovis/Streptococcus equinus complex are consumed by millions of people daily and thus may be safe.[28] 25 to 80% of people with Streptococcus bovis/gallolyticus bacteremia have concomitant colorectal tumors.[29] Seroprevalence of Streptococcus bovis/gallolyticus is considered as a candidate practical marker for the early prediction of an underlying bowel lesion at high risk population.[29] It has been suggested that the presence of antibodies to Streptococcus bovis/gallolyticus antigens or the antigens themselves in the bloodstream may act as markers for the carcinogenesis in the colon.[29]
Pathogenic Escherichia coli may increase the risk of colorectal cancer by producing the genotoxic metabolite, colibactin.[30]
Inflammatory bowel disease
[edit]People with inflammatory bowel disease (ulcerative colitis and Crohn's disease) are at increased risk of colon cancer.[31][32] The risk increases the longer a person has the disease, and the worse the severity of inflammation.[33] In these high risk groups, both prevention with aspirin and regular colonoscopies are recommended.[34] Endoscopic surveillance in this high-risk population may reduce the development of colorectal cancer through early diagnosis and may also reduce the chances of dying from colon cancer.[34] People with inflammatory bowel disease account for less than 2% of colon cancer cases yearly.[33] In those with Crohn's disease, 2% get colorectal cancer after 10 years, 8% after 20 years, and 18% after 30 years.[33] In people who have ulcerative colitis, approximately 16% develop either a cancer precursor or cancer of the colon over 30 years.[33]
Genetics
[edit]Those with a family history in two or more first-degree relatives (such as a parent or sibling) have a two to threefold greater risk of disease, and this group accounts for about 20% of all cases. A number of genetic syndromes are also associated with higher rates of colorectal cancer. The most common of these is hereditary nonpolyposis colorectal cancer (HNPCC, or Lynch syndrome) which is present in about 3% of people with colorectal cancer.[20] Other syndromes that are strongly associated with colorectal cancer include Gardner syndrome and familial adenomatous polyposis (FAP).[35] For people with these syndromes, cancer almost always occurs and makes up 1% of the cancer cases.[36] A total proctocolectomy may be recommended for people with FAP as a preventive measure due to the high risk of malignancy. Colectomy, removal of the colon, may not suffice as a preventive measure because of the high risk of rectal cancer if the rectum remains.[37] The most common polyposis syndrome affecting the colon is serrated polyposis syndrome,[38] which is associated with a 25-40% risk of CRC.[39]
Mutations in the pair of genes (POLE and POLD1) have been associated with familial colon cancer.[40]
Most deaths due to colon cancer are associated with metastatic disease. A gene that appears to contribute to the potential for metastatic disease, metastasis associated in colon cancer 1 (MACC1), has been isolated.[41] It is a transcriptional factor that influences the expression of hepatocyte growth factor. This gene is associated with the proliferation, invasion, and scattering of colon cancer cells in cell culture, and tumor growth and metastasis in mice. MACC1 may be a potential target for cancer intervention, but this possibility needs to be confirmed with clinical studies.[42]
Epigenetic factors, such as abnormal DNA methylation of tumor suppressor promoters, play a role in the development of colorectal cancer.[43]
Ashkenazi Jews have a 6% higher risk rate of getting adenomas and then colon cancer due to mutations in the APC gene being more common.[44]
Pathogenesis
[edit]Colorectal cancer is a disease originating from the epithelial cells lining the colon or rectum of the gastrointestinal tract, most frequently as a result of genetic mutations in the Wnt signaling pathway that increases signaling activity.[45] The Wnt signaling pathway normally plays an important role for normal function of these cells including maintaining this lining. Mutations can be inherited or acquired, and most probably occur in the intestinal crypt stem cell.[46][47][48] The most commonly mutated gene in all colorectal cancer is the APC gene, which produces the APC protein.[45] The APC protein prevents the accumulation of β-catenin protein. Without APC, β-catenin accumulates to high levels and translocates (moves) into the nucleus, binds to DNA, and activates the transcription of proto-oncogenes. These genes are normally important for stem cell renewal and differentiation, but when inappropriately expressed at high levels, they can cause cancer.[45] While APC is mutated in most colon cancers, some cancers have increased β-catenin because of mutations in β-catenin (CTNNB1) that block its own breakdown, or have mutations in other genes with function similar to APC such as AXIN1, AXIN2, TCF7L2, or NKD1.[49]
Beyond the defects in the Wnt signaling pathway, other mutations must occur for the cell to become cancerous. The p53 protein, produced by the TP53 gene, normally monitors cell division and induces their programmed death if they have Wnt pathway defects. Eventually, a cell line acquires a mutation in the TP53 gene and transforms the tissue from a benign epithelial tumor into an invasive epithelial cell cancer. Sometimes the gene encoding p53 is not mutated, but another protective protein named BAX is mutated instead.[49]
Other proteins responsible for programmed cell death that are commonly deactivated in colorectal cancers are TGF-β and DCC (Deleted in Colorectal Cancer). TGF-β has a deactivating mutation in at least half of colorectal cancers. Sometimes TGF-β is not deactivated, but a downstream protein named SMAD is deactivated.[49] DCC commonly has a deleted segment of a chromosome in colorectal cancer.[50]
Approximately 70% of all human genes are expressed in colorectal cancer, with just over 1% of having increased expression in colorectal cancer compared to other forms of cancer.[51] Some genes are oncogenes: they are overexpressed in colorectal cancer. For example, genes encoding the proteins KRAS, RAF, and PI3K, which normally stimulate the cell to divide in response to growth factors, can acquire mutations that result in over-activation of cell proliferation. The chronological order of mutations is sometimes important. If a previous APC mutation occurred, a primary KRAS mutation often progresses to cancer rather than a self-limiting hyperplastic or borderline lesion.[52] PTEN, a tumor suppressor, normally inhibits PI3K, but can sometimes become mutated and deactivated.[49]
Comprehensive, genome-scale analysis has revealed that colorectal carcinomas can be categorized into hypermutated and non-hypermutated tumor types.[53] In addition to the oncogenic and inactivating mutations described for the genes above, non-hypermutated samples also contain mutated CTNNB1, FAM123B, SOX9, ATM, and ARID1A. Progressing through a distinct set of genetic events, hypermutated tumors display mutated forms of ACVR2A, TGFBR2, MSH3, MSH6, SLC9A9, TCF7L2, and BRAF. The common theme among these genes, across both tumor types, is their involvement in Wnt and TGF-β signaling pathways, which results in increased activity of MYC, a central player in colorectal cancer.[53]
Mismatch repair (MMR) deficient tumours are characterized by a relatively high number of poly-nucleotide tandem repeats.[54] This is caused by a deficiency in MMR proteins – which are typically caused by epigenetic silencing and or inherited mutations (e.g., Lynch syndrome).[55] 15 to 18 percent of colorectal cancer tumours have MMR deficiencies, with 3 percent developing due to Lynch syndrome.[56] The role of the mismatch repair system is to protect the integrity of the genetic material within cells (i.e., error detecting and correcting).[55] Consequently, a deficiency in MMR proteins may lead to an inability to detect and repair genetic damage, allowing for further cancer-causing mutations to occur and colorectal cancer to progress.[55]
The polyp to cancer progression sequence is the classical model of colorectal cancer pathogenesis.[57] In this adenoma-carcinoma sequence,[58] normal epithelial cells progress to dysplastic cells such as adenomas, and then to carcinoma, by a process of progressive genetic mutation.[59] Central to the polyp to CRC sequence are gene mutations, epigenetic alterations, and local inflammatory changes.[57] The polyp to CRC sequence can be used as an underlying framework to illustrate how specific molecular changes lead to various cancer subtypes.[57]
Field defects
[edit]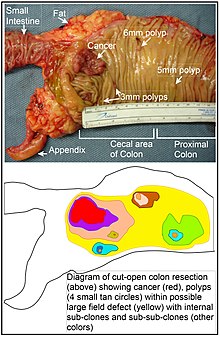
The term "field cancerization" was first used in 1953 to describe an area or "field" of epithelium that has been preconditioned (by what were largely unknown processes at the time) to predispose it towards development of cancer.[60] Since then, the terms "field cancerization", "field carcinogenesis", "field defect", and "field effect" have been used to describe pre-malignant or pre-neoplastic tissue in which new cancers are likely to arise.[61]
Field defects are important in progression to colon cancer.[62][63]
However, as pointed out by Rubin, "The vast majority of studies in cancer research has been done on well-defined tumors in vivo, or on discrete neoplastic foci in vitro. Yet there is evidence that more than 80% of the somatic mutations found in mutator phenotype human colorectal tumors occur before the onset of terminal clonal expansion."[64][65] Similarly, Vogelstein et al.[66] pointed out that more than half of somatic mutations identified in tumors occurred in a pre-neoplastic phase (in a field defect), during growth of apparently normal cells. Likewise, epigenetic alterations present in tumors may have occurred in pre-neoplastic field defects.[67]
An expanded view of field effect has been termed "etiologic field effect", which encompasses not only molecular and pathologic changes in pre-neoplastic cells but also influences of exogenous environmental factors and molecular changes in the local microenvironment on neoplastic evolution from tumor initiation to death.[68]
Epigenetics
[edit]Epigenetic alterations are much more frequent in colon cancer than genetic (mutational) alterations. As described by Vogelstein et al.,[66] an average cancer of the colon has only 1 or 2 oncogene mutations and 1 to 5 tumor suppressor mutations (together designated "driver mutations"), with about 60 further "passenger" mutations. The oncogenes and tumor suppressor genes are well studied and are described above under Pathogenesis.[69][70]
In addition to epigenetic alteration of expression of miRNAs, other common types of epigenetic alterations in cancers that change gene expression levels include direct hypermethylation or hypomethylation of CpG islands of protein-encoding genes and alterations in histones and chromosomal architecture that influence gene expression.[71] As an example, 147 hypermethylations and 27 hypomethylations of protein coding genes were frequently associated with colorectal cancers. Of the hypermethylated genes, 10 were hypermethylated in 100% of colon cancers, and many others were hypermethylated in more than 50% of colon cancers.[72] In addition, 11 hypermethylations and 96 hypomethylations of miRNAs were also associated with colorectal cancers.[72] Abnormal (aberrant) methylation occurs as a normal consequence of normal aging and the risk of colorectal cancer increases as a person gets older.[73] The source and trigger of this age-related methylation is unknown.[73][74] Approximately half of the genes that show age-related methylation changes are the same genes that have been identified to be involved in the development of colorectal cancer.[73] These findings may suggest a reason for age being associated with the increased risk of developing colorectal cancer.[73]
Epigenetic reductions of DNA repair enzyme expression may likely lead to the genomic and epigenomic instability characteristic of cancer.[75][76][67] As summarized in the articles Carcinogenesis and Neoplasm, for sporadic cancers in general, a deficiency in DNA repair is occasionally due to a mutation in a DNA repair gene, but is much more frequently due to epigenetic alterations that reduce or silence expression of DNA repair genes.[77]
Epigenetic alterations involved in the development of colorectal cancer may affect a person's response to chemotherapy.[78]
Genomics and epigenomics
[edit]Consensus molecular subtypes (CMS) classification of colorectal cancer was first introduced in 2015. CMS classification so far has been considered the most robust classification system available for CRC that has a clear biological interpretability and the basis for future clinical stratification and subtype-based targeted interventions.[79]
A novel Epigenome-based Classification (EpiC) of colorectal cancer was proposed in 2021 introducing 4 enhancer subtypes in people with CRC. Chromatin states using 6 histone marks are characterized to identify EpiC subtypes. A combinatorial therapeutic approach based on the previously introduced consensus molecular subtypes (CMSs) and EpiCs could significantly enhance current treatment strategies.[80]
Diagnosis
[edit]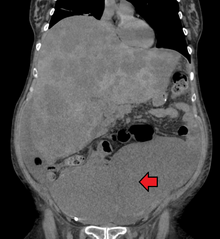
Colorectal cancer diagnosis is performed by sampling of areas of the colon suspicious for possible tumor development, typically during colonoscopy or sigmoidoscopy, depending on the location of the lesion.[20]
Medical imaging
[edit]A colorectal cancer is sometimes initially discovered on CT scan.[81]
Presence of metastases is determined by a CT scan of the chest, abdomen and pelvis.[20] Other potential imaging tests such as PET and MRI may be used in certain cases.[20] MRI is particularly useful to determine local stage of the tumor and to plan the optimal surgical approach.[81]
MRI is also performed after completion of neoadjuvant chemoradiotherapy to identify patients who achieve complete response. Patients with complete response on both MRI and endoscopy may not require surgical resection and can avoid unnecessary surgical morbidity and complications.[82] Patients selected for non-surgical treatment of rectal cancer should have periodic MRI scans, receive physical examinations, and undergo endoscopy procedures to detect any tumor re-growth which can occur in a minority of these patients. When local recurrence occurs, periodic follow up can detect it when it is still small and curable with salvage surgery. In addition, MRI tumor regression grades can be assigned after chemoradiotherapy which correlate with patients' long-term survival outcomes.[83]
Histopathology
[edit]

The histopathologic characteristics of the tumor are reported from the analysis of tissue taken from a biopsy or surgery. A pathology report contains a description of the microscopical characteristics of the tumor tissue, including both tumor cells and how the tumor invades into healthy tissues and finally if the tumor appears to be completely removed. The most common form of colon cancer is adenocarcinoma, constituting between 95%[85] and 98%[86] of all cases of colorectal cancer. Other, rarer types include lymphoma, adenosquamous and squamous cell carcinoma. Some subtypes are more aggressive.[87] Immunohistochemistry may be used in uncertain cases.[88]
Staging
[edit]Staging of the cancer is based on both radiological and pathological findings. As with most other forms of cancer, tumor staging is based on the TNM system which considers how much the initial tumor has spread and the presence of metastases in lymph nodes and more distant organs.[20] The AJCC 8th edition was published in 2018.[89]
Prevention
[edit]It has been estimated that about half of colorectal cancer cases are due to lifestyle factors, and about a quarter of all cases are preventable.[90] Increasing surveillance, engaging in physical activity, consuming a diet high in fiber, quitting smoking and limiting alcohol consumption decrease the risk.[91][92]
Lifestyle
[edit]Lifestyle risk factors with strong evidence include lack of exercise, cigarette smoking, alcohol, and obesity.[93][94][95] The risk of colon cancer can be reduced by maintaining a normal body weight through a combination of sufficient exercise and eating a healthy diet.[96]
Current research consistently links eating more red meat and processed meat to a higher risk of the disease.[97] Starting in the 1970s, dietary recommendations to prevent colorectal cancer often included increasing the consumption of whole grains, fruits and vegetables, and reducing the intake of red meat and processed meats. This was based on animal studies and retrospective observational studies. However, large scale prospective studies have failed to demonstrate a significant protective effect, and due to the multiple causes of cancer and the complexity of studying correlations between diet and health, it is uncertain whether any specific dietary interventions will have significant protective effects.[98]: 432–433 [99]: 125–126 In 2018 the National Cancer Institute stated that "There is no reliable evidence that a diet started in adulthood that is low in fat and meat and high in fiber, fruits, and vegetables reduces the risk of CRC by a clinically important degree."[93][100]
Consuming alcoholic drinks and consuming processed meat both increase the risk of colorectal cancer.[101]
The 2014 World Health Organization cancer report noted that it has been hypothesized that dietary fiber might help prevent colorectal cancer, but that most studies at the time had not yet studied the correlation.[99] A 2019 review, however, found evidence of benefit from dietary fiber and whole grains.[102] The World Cancer Research Fund listed the benefit of fiber for prevention of colorectal cancer as "probable" as of 2017.[103] A 2022 umbrella review says there is "convincing evidence" for that association.[104]
Higher physical activity is recommended.[21][105] Physical exercise is associated with a modest reduction in colon but not rectal cancer risk.[106][107] High levels of physical activity reduce the risk of colon cancer by about 21%.[108] Sitting regularly for prolonged periods is associated with higher mortality from colon cancer. Regular exercise does not negate the risk but does lower it.[109]
Medication and supplements
[edit]Aspirin and celecoxib appear to decrease the risk of colorectal cancer in those at high risk.[110][111] Aspirin is recommended in those who are 50 to 60 years old, do not have an increased risk of bleeding, and are at risk for cardiovascular disease to prevent colorectal cancer.[112] It is not recommended in those at average risk.[113]
There is tentative evidence for calcium supplementation, but it is not sufficient to make a recommendation.[114]
Adequate Vitamin D intake and blood levels are associated with a lower risk of colon cancer.[115][116]
Screening
[edit]As more than 80% of colorectal cancers arise from adenomatous polyps, screening for this cancer is effective for both early detection and for prevention.[20][117] Diagnosis of cases of colorectal cancer through screening tends to occur 2–3 years before diagnosis of cases with symptoms.[20] Any polyps that are detected can be removed, usually by colonoscopy or sigmoidoscopy, and thus prevent them from turning into cancer. Screening has the potential to reduce colorectal cancer deaths by 60%.[118]
The three main screening tests are colonoscopy, fecal occult blood testing, and flexible sigmoidoscopy. Of the three, only sigmoidoscopy cannot screen the right side of the colon where 42% of cancers are found.[119] Flexible sigmoidoscopy, however, has the best evidence for decreasing the risk of death from any cause.[120]
Fecal occult blood testing (FOBT) of the stool is typically recommended every two years and can be either guaiac-based or immunochemical.[20] If abnormal FOBT results are found, participants are typically referred for a follow-up colonoscopy examination. When done once every 1–2 years, FOBT screening reduces colorectal cancer deaths by 16% and among those participating in screening, colorectal cancer deaths can be reduced up to 23%, although it has not been proven to reduce all-cause mortality.[121] Immunochemical tests are accurate and do not require dietary or medication changes before testing.[122] However, research in the UK has found that for these immunochemical tests, the threshold for further investigation is set at a point that may miss more than half of bowel cancer cases. The research suggests that the NHS England's Bowel Cancer Screening Programme could make better use of the test's ability to provide the exact concentration of blood in faeces (rather than only whether it is above or below a cutoff level).[123][124]
Other options include virtual colonoscopy and stool DNA screening testing (FIT-DNA). Virtual colonoscopy via a CT scan appears as good as standard colonoscopy for detecting cancers and large adenomas but is expensive, associated with radiation exposure, and cannot remove any detected abnormal growths as standard colonoscopy can.[20] Stool DNA screening test looks for biomarkers associated with colorectal cancer and precancerous lesions, including altered DNA and blood hemoglobin. A positive result should be followed by colonoscopy. FIT-DNA has more false positives than FIT and thus results in more adverse effects.[10] Further study is required as of 2016 to determine whether a three-year screening interval is correct.[10]
Recommendations
[edit]In the United States, screening is typically recommended between ages 50 and 75 years.[10][125] The American Cancer Society recommends starting at the age of 45.[126] For those between 76 and 85 years old, the decision to screen should be individualized.[10] For those at high risk, screenings usually begin at around 40.[20][127]
Several screening methods are recommended including stool-based tests every 2 years, sigmoidoscopy every 10 years with fecal immunochemical testing every two years, and colonoscopy every 10 years.[125] It is unclear which of these two methods is better.[128] Colonoscopy may find more cancers in the first part of the colon, but is associated with greater cost and more complications.[128] For people with average risk who have had a high-quality colonoscopy with normal results, the American Gastroenterological Association does not recommend any type of screening in the 10 years following the colonoscopy.[129][130] For people over 75 or those with a life expectancy of less than 10 years, screening is not recommended.[131] It takes about 10 years after screening for one out of a 1000 people to benefit.[132] The USPSTF list seven potential strategies for screening, with the most important thing being that at least one of these strategies is appropriately used.[10]
In Canada, among those 50 to 75 years old at normal risk, fecal immunochemical testing or FOBT is recommended every two years or sigmoidoscopy every 10 years.[133] Colonoscopy is less preferred.[133]
Some countries have national colorectal screening programs which offer FOBT screening for all adults within a certain age group, typically starting between ages 50 and 60. Examples of countries with organised screening include the United Kingdom,[134] Australia,[135] the Netherlands,[136] Hong Kong, and Taiwan.[137]
The UK Bowel Cancer Screening Programme aims to find warning signs in people aged 60 to 74, by recommending a faecal immunochemical test (FIT) every two years. FIT measures blood in faeces, and people with levels above a certain threshold may have bowel tissue examined for signs of cancer. Growths having cancerous potential are removed.[138][124]
Treatment
[edit]The treatment of colorectal cancer can be aimed at cure or palliation. The decision on which aim to adopt depends on various factors, including the person's health and preferences, as well as the stage of the tumor.[139] Assessment in multidisciplinary teams is a critical part of determining whether the patient is suitable for surgery or not.[140] When colorectal cancer is caught early, surgery can be curative. However, when it is detected at later stages (for which metastases are present), this is less likely and treatment is often directed at palliation, to relieve symptoms caused by the tumour and keep the person as comfortable as possible.[20]
Surgery
[edit]


At an early stage, colorectal cancer may be removed during a colonoscopy using one of several techniques, including endoscopic mucosal resection or endoscopic submucosal dissection.[5] Endoscopic resection is possible if there is low possibility of lymph node metastasis and the size and location of the tumor make en bloc resection possible.[141] For people with localized cancer, the preferred treatment is complete surgical removal with adequate margins, with the attempt of achieving a cure. The procedure of choice is a partial colectomy (or proctocolectomy for rectal lesions) where the affected part of the colon or rectum is removed along with parts of its mesocolon and blood supply to facilitate removal of draining lymph nodes. This can be done either by an open laparotomy or laparoscopically, depending on factors related to the individual person and lesion factors.[20] The colon may then be reconnected or a person may have a colostomy.[5]
If there are only a few metastases in the liver or lungs, these may also be removed. Chemotherapy may be used before surgery to shrink the cancer before attempting to remove it. The two most common sites of recurrence of colorectal cancer are the liver and lungs.[20] For peritoneal carcinomatosis cytoreductive surgery, sometimes in combination with HIPEC can be used in an attempt to remove the cancer.[142]
Chemotherapy
[edit]In both cancer of the colon and rectum, chemotherapy may be used in addition to surgery in certain cases. The decision to add chemotherapy in management of colon and rectal cancer depends on the stage of the disease.[143]
In Stage I colon cancer, no chemotherapy is offered, and surgery is the definitive treatment. The role of chemotherapy in Stage II colon cancer is debatable, and is usually not offered unless risk factors such as T4 tumor, undifferentiated tumor, vascular and perineural invasion or inadequate lymph node sampling is identified.[144] It is also known that the people who carry abnormalities of the mismatch repair genes do not benefit from chemotherapy. For Stage III and Stage IV colon cancer, chemotherapy is an integral part of treatment.[20]
If cancer has spread to the lymph nodes or distant organs, which is the case with Stage III and Stage IV colon cancer respectively, adding chemotherapy agents fluorouracil, capecitabine or oxaliplatin increases life expectancy. If the lymph nodes do not contain cancer, the benefits of chemotherapy are controversial. If the cancer is widely metastatic or unresectable, treatment is then palliative. Typically in this setting, a number of different chemotherapy medications may be used.[20] Chemotherapy drugs for this condition may include capecitabine, fluorouracil, irinotecan, oxaliplatin and UFT.[145] The drugs capecitabine and fluorouracil are interchangeable, with capecitabine being an oral medication and fluorouracil being an intravenous medicine. Some specific regimens used for CRC are CAPOX, FOLFOX, FOLFOXIRI, and FOLFIRI.[146] Antiangiogenic drugs such as bevacizumab are often added in first line therapy.[147] Another class of drugs used in the second line setting are epidermal growth factor receptor inhibitors, of which the three FDA approved ones are aflibercept, cetuximab and panitumumab.[148][149]
The primary difference in the approach to low stage rectal cancer is the incorporation of radiation therapy. Often, it is used in conjunction with chemotherapy in a neoadjuvant fashion to enable surgical resection, so that ultimately a colostomy is not required. However, it may not be possible in low lying tumors, in which case, a permanent colostomy may be required. Stage IV rectal cancer is treated similar to Stage IV colon cancer.
Stage IV colorectal cancer due to peritoneal carcinomatosis can be treated using HIPEC combined with cytoreductive surgery, in some people.[150][151][152] Also, T4 colorectal cancer can be treated with HIPEC to avoid future relapses.[153]
Radiation therapy
[edit]While a combination of radiation and chemotherapy may be useful for rectal cancer,[20] for some people requiring treatment, chemoradiotherapy can increase acute treatment-related toxicity, and has not been shown to improve survival rates compared to radiotherapy alone, although it is associated with less local recurrence.[142] For squamous cell carcinoma of the anal canal, chemoradiation therapy (CRT) with 5-FU and mitomycin C is preferred over radiation alone, offering improved survival outcomes but with increased risks of acute hematological toxicity.[154]
The use of radiotherapy in colon cancer is not routine due to the sensitivity of the bowels to radiation.[155] Radiation therapy's side effects (and occurrence rates) include acute (27%) and late (17%) dermatological toxicities, acute (14%) and late (27%) gastrointestinal toxicities,[154] and late pelvic radiation disease (1-10%), e.g., irreversible lumbosacral plexopathy.[156]
As with chemotherapy, radiotherapy can be used as a neoadjuvant for clinical stages T3 and T4 for rectal cancer.[157] This results in downsizing or downstaging of the tumour, preparing it for surgical resection, and also decreases local recurrence rates.[157] For locally advanced rectal cancer, neoadjuvant chemoradiotherapy has become the standard treatment.[158] Additionally, when surgery is not possible radiation therapy has been suggested to be an effective treatment against CRC pulmonary metastases, which are developed by 10-15% of people with CRC.[159]
Immunotherapy
[edit]Immunotherapy with immune checkpoint inhibitors has been found to be useful for a type of colorectal cancer with mismatch repair deficiency and microsatellite instability.[160][161][162] Pembrolizumab is approved for advanced CRC tumours that are MMR deficient and have failed usual treatments.[163] Most people who do improve, however, still worsen after months or years.[161]
On the other hand, in a prospective phase 2 study published in June 2022 in The New England Journal of Medicine, 12 patients with Deficient Mismatch Repair (dMMR) stage II or III rectal adenocarcinoma were administered single-agent dostarlimab, an anti–PD-1 monoclonal antibody, every three weeks for six months. After a median follow-up of 12 months (range, 6 to 25 months), all 12 patients had a complete clinical response with no evidence of tumor on MRI, 18F-fluorodeoxyglucose–positron-emission tomography, endoscopic evaluation, digital rectal examination, or biopsy. Moreover, no patient in the trial needed chemoradiotherapy or surgery, and no patient reported adverse events of grade 3 or higher. However, although the results of this study are promising, the study is small and has uncertainties about long-term outcomes.[164]
Palliative care
[edit]Palliative care can be used at the same time as the cancer treatment and is recommended for any person who has advanced colon cancer or who has significant symptoms.[165][166] Involvement of palliative care may be beneficial to improve the quality of life for both the person and his or her family, by improving symptoms, anxiety and preventing admissions to the hospital.[167]
In people with incurable colorectal cancer, palliative care can consist of procedures that relieve symptoms or complications from the cancer but do not attempt to cure the underlying cancer, thereby improving quality of life. Surgical options may include non-curative surgical removal of some of the cancer tissue, bypassing part of the intestines, or stent placement. These procedures can be considered to improve symptoms and reduce complications such as bleeding from the tumor, abdominal pain and intestinal obstruction.[168] Non-operative methods of symptomatic treatment include radiation therapy to decrease tumor size as well as pain medications.[169]
Psychosocial Intervention
[edit]In addition to medical intervention, a variety of psychosocial interventions have been implemented to address psychosocial concerns in the context of colorectal cancer.[170] Depression and anxiety are highly prevalent in patients diagnosed with CRC, therefore psychosocial interventions can be helpful for alleviating psychological distress.[171][172] Many patients continue to experience symptoms of anxiety and depression following treatment, regardless of treatment outcome.[171][173] Societal stigmas associated with colorectal cancer present further psychosocial challenges for CRC patients and their families.[174][175]
Depression and Anxiety
[edit]Colorectal cancer patients have a 51% higher risk of experiencing depression than individuals without the disease.[172] Additionally, CRC patients are at high risk of experiencing severe anxiety, low self-esteem, poor self-concept, and social anxiety.[171][176]
Post-Treatment Distress
[edit]Regardless of treatment outcome, many CRC patients experience ongoing symptoms of anxiety, depression, and distress.[171]
Survivorship of CRC can involve significant lifestyle adjustments.[175] Postoperative afflictions may include stomas, bowel issues, incontinence, odor, and changes to sexual functioning.[175][176] These changes can result in distorted body image, social anxiety, depression, and distress—all of which contribute to a poorer quality of life.[175][177]
Colorectal cancer is the second leading cause of cancer-related death worldwide.[178] Transitioning into palliative care and contending with mortality can be a deeply distressing experience for a CRC patient and their loved ones.
Stigma
[edit]Colorectal cancer is highly stigmatized and can elicit feelings of disgust from patients, healthcare professionals, family, intimate partners, and the general public.[174] Patients with stomas are especially vulnerable to stigmatization due to unavoidable odors, gas, and unpleasant noises from stoma bags.[174] Additionally, associated CRC risk factors like poor diet, alcohol consumption, and lack of physical activity prompt negative assumptions of blame and personal responsibility onto CRC patients.[175] Judgement from others along with internalized self-blame and embarrassment can negatively affect self-esteem, sociability, and quality of life.[175]
Methods of Intervention
[edit]Face-to-face interventions such as clinician-patient talk therapy, body-mind-spirit practices, and support group sessions have been identified as most effective in reducing anxiety and depression in CRC patients.[170] Additionally, journaling exercises and over-the-phone talk therapy sessions have been implemented.[170] Though deemed less effective, these non-face-to-face interventions are economically inclusive and have been found to reduce both depression and anxiety in CRC patients.[170]
Follow-up
[edit]The U.S. National Comprehensive Cancer Network and American Society of Clinical Oncology provide guidelines for the follow-up of colon cancer.[179][180] A medical history and physical examination are recommended every 3 to 6 months for 2 years, then every 6 months for 5 years. Carcinoembryonic antigen blood level measurements follow the same timing, but are only advised for people with T2 or greater lesions who are candidates for intervention. A CT-scan of the chest, abdomen and pelvis can be considered annually for the first 3 years for people who are at high risk of recurrence (for example, those who had poorly differentiated tumors or venous or lymphatic invasion) and are candidates for curative surgery (with the aim to cure). A colonoscopy can be done after 1 year, except if it could not be done during the initial staging because of an obstructing mass, in which case it should be performed after 3 to 6 months. If a villous polyp, a polyp >1 centimeter or high-grade dysplasia is found, it can be repeated after 3 years, then every 5 years. For other abnormalities, the colonoscopy can be repeated after 1 year.[143]
Routine PET or ultrasound scanning, chest X-rays, complete blood count or liver function tests are not recommended.[179][180]
For people who have undergone curative surgery or adjuvant therapy (or both) to treat non-metastatic colorectal cancer, intense surveillance and close follow-up have not been shown to provide additional survival benefits.[181]
Exercise
[edit]Exercise may be recommended in the future as secondary therapy to cancer survivors. In epidemiological studies, exercise may decrease colorectal cancer-specific mortality and all-cause mortality. Results for the specific amounts of exercise needed to observe a benefit were conflicting. These differences may reflect differences in tumour biology and the expression of biomarkers. People with tumors that lacked CTNNB1 expression (β-catenin), involved in Wnt signalling pathway, required more than 18 Metabolic equivalent (MET) hours per week, a measure of exercise, to observe a reduction in colorectal cancer mortality. The mechanism of how exercise benefits survival may be involved in immune surveillance and inflammation pathways. In clinical studies, a pro-inflammatory response was found in people with stage II-III colorectal cancer who underwent 2 weeks of moderate exercise after completing their primary therapy. Oxidative balance may be another possible mechanism for benefits observed. A significant decrease in 8-oxo-dG was found in the urine of people who underwent 2 weeks of moderate exercise after primary therapy. Other possible mechanisms may involve metabolic hormone and sex-steroid hormones, although these pathways may be involved in other types of cancers.[182][183]
Another potential biomarker may be p27. Survivors with tumors that expressed p27 and performed greater and equal to 18 MET hours per week were found to have reduced colorectal cancer mortality survival compared to those with less than 18 MET hours per week. Survivors without p27 expression who exercised were shown to have worse outcomes. The constitutive activation of PI3K/AKT/mTOR pathway may explain the loss of p27 and excess energy balance may up-regulate p27 to stop cancer cells from dividing.[183]
Physical activity provides benefits to people with non-advanced colorectal cancer. Improvements in aerobic fitness, cancer-related fatigue and health-related quality of life have been reported in the short term.[184] However, these improvements were not observed at the level of disease-related mental health, such as anxiety and depression.[184]
Prognosis
[edit]Fewer than 600 genes are linked to outcomes in colorectal cancer.[51] These include both unfavorable genes, where high expression is related to poor outcome, for example the heat shock 70 kDa protein 1 (HSPA1A), and favorable genes where high expression is associated with better survival, for example the putative RNA-binding protein 3 (RBM3).[51] The prognosis is also correlated with a poor fidelity of the pre-mRNA splicing apparatus, and thus a high number of deviating alternative splicing.[185]
Recurrence rates
[edit]The average five-year recurrence rate in people with colon cancer where surgery is successful is 5% for stage I cancers, 12% in stage II and 33% in stage III. However, depending on the number of risk factors it ranges from 9–22% in stage II and 17–44% in stage III.[186] The average five-year recurrence rate in people with rectal cancer where surgery is successful is 9% for stage 0 (after pre-treatment) cancers, 8% for stage I cancers, 18% in stage II and 34% in stage III. Depending on the number of risk factors (0-2) the risk for distant metastasis in rectal cancer ranges from 4-11% in stage 0, 6-12% in stage I, 11-28% in stage II and 15-43% in stage III.[187]
The recurrence rates have decreased over the past decades as a result of improvements in the colorectal cancer management.[188] The risk of recurrence after five years of surveillance remain very low.[189]
Survival rates
[edit]In Europe the five-year survival rate for colorectal cancer is less than 60%. In the developed world about a third of people who get the disease die from it.[20]
Survival is directly related to detection and the type of cancer involved, but overall is poor for symptomatic cancers, as they are typically quite advanced. Survival rates for early stage detection are about five times that of late stage cancers. People with a tumor that has not breached the muscularis mucosa (TNM stage Tis, N0, M0) have a five-year survival rate of 100%, while those with invasive cancer of T1 (within the submucosal layer) or T2 (within the muscular layer) have an average five-year survival rate of approximately 90%. Those with a more invasive tumor yet without node involvement (T3-4, N0, M0) have an average five-year survival rate of approximately 70%. People with positive regional lymph nodes (any T, N1-3, M0) have an average five-year survival rate of approximately 40%, while those with distant metastases (any T, any N, M1) have a poor prognosis and the five year survival ranges from <5 percent to 31 percent.[190][191][192][193][194]
Five-year overall survival (OS) in rectal cancer after modern preoperative treatment and surgery was 90% for stage 0, 86% for stage I, 78% for stage II, and 67% for stage III according to a nationwide, population-based study.[187]
Whilst the impact of colorectal cancer on those who survive varies greatly there will often be a need to adapt to both physical and psychological outcomes of the illness and its treatment.[195] For example, it is common for people to experience incontinence,[196] sexual dysfunction,[197] problems with stoma care[198] and fear of cancer recurrence[199] after primary treatment has concluded.
A qualitative systematic review published in 2021 highlighted that there are three main factors influencing adaptation to living with and beyond colorectal cancer: support mechanisms, severity of late effects of treatment and psychosocial adjustment. Therefore, it is essential that people are offered appropriate support to help them better adapt to life following treatment.[200]
Epidemiology
[edit]
Globally more than 1 million people get colorectal cancer every year[20] resulting in about 715,000 deaths as of 2010 up from 490,000 in 1990.[201]
As of 2012[update], it is the second most common cause of cancer in women (9.2% of diagnoses) and the third most common in men (10.0%)[14]: 16 with it being the fourth most common cause of cancer death after lung, stomach, and liver cancer.[202] It is more common in developed than developing countries.[203] Global incidence varies 10-fold, with highest rates in Australia, New Zealand, Europe and the US and lowest rates in Africa and South-Central Asia.[204]
United States
[edit]In 2022, the incidence of colorectal cancer in the United States was anticipated to be about 151,000 adults, including over 106,000 new cases of colon cancer (some 54,000 men and 52,000 women) and about 45,000 new cases of rectal cancer.[205] Since the 1980s, the incidence of colorectal cancer decreased, dropping by about 2% annually from 2014 to 2018 in adults aged 50 and older, due mainly to improved screening.[205] However, incidence of colorectal cancer has increased in individuals aged 25 to 50. In early 2023, the American Cancer Society (ACS) reported that 20% of diagnoses (of colon cancer) in 2019 were in patients under age 55, which is about double the rate in 1995, and rates of advanced disease increased by about 3% annually in people younger than 50. It predicted that, in 2023, an estimated 19,550 diagnoses and 3,750 deaths would be in people younger than 50.[206] Colorectal cancer also disproportionately affects the Black community, where the rates are the highest of any racial/ethnic group in the US. African Americans are about 20% more likely to get colorectal cancer and about 40% more likely to die from it than most other groups. Black Americans often experience greater obstacles to cancer prevention, detection, treatment, and survival, including systemic racial disparities that are complex and go beyond the obvious connection to cancer.
United Kingdom
[edit]In the UK about 41,000 people a year get colon cancer making it the fourth most common type.[207]
Australia
[edit]One in 19 men and one in 28 women in Australia will develop colorectal cancer before the age of 75; one in 10 men and one in 15 women will develop it by 85 years of age.[208]
Papua New Guinea
[edit]In Papua New Guinea and other Pacific Island States including the Solomon Islands, colorectal cancer is a very rare cancer compared to lung, stomach, liver or breast cancer. It is estimated that 8 in 100,000 people are likely to develop colorectal cancer every year, while 24 in 100,000 women are likely to develop breast cancer.[209]
Early-onset colorectal cancer (EOCC)
[edit]A diagnosis of colorectal cancer in patients under 50 years of age is referred to as early-onset colorectal cancer (EOCC).[178][210] Instances of EOCC have increased over the last decade, specifically in patient populations aged 20 to 40 years old throughout North America, Europe, Australia, and China.[210][211]
Incidence by age
[edit]The incidence of colorectal cancer in younger populations has increased over the last decade.[178][210][211] While advancements in diagnostic procedure may have some impact, reduced likelihood of screening among these populations suggests detection bias is not a major contributor to this trend. It is more likely that cohort effects are contributing.[211]
The population experiencing the greatest rise in EOCC cases are men and women aged 20 to 29 years old, with incidence increasing by 7.9% per year between 2004 and 2016.[211] Similarly, though less severe, men and women aged 30 to 39 experienced an increase in cases at a rate of 3.4% per year during that same time period. Despite these increases, the mortality rate for colorectal cancer has remained the same.[211]
Risk factors
[edit]Risk factors associated with EOCC are akin to those of all colorectal cancer cases.[210] Observed cohort-effects are likely the product of generational shifts in lifestyle and environmental factors.[178][210]
Preventative screening
[edit]In 2018, the American Cancer Society modified their previous screening guideline for colorectal cancer from age 50 down to age 45 following the recognition of increasing cases of EOCC.[211] Individuals under the age of 60 have been identified as most susceptible to non-participation in colorectal cancer screening.[212]
History
[edit]This section needs expansion. You can help by adding to it. (January 2024) |
Rectal cancer has been diagnosed in an Ancient Egyptian mummy who had lived in the Dakhleh Oasis during the Ptolemaic period.[213]
Society and culture
[edit]In the United States, March is colorectal cancer awareness month.[118]
Research
[edit]This section needs to be updated. The reason given is: all sources > 5 years. (January 2024) |
Preliminary in-vitro evidence suggests lactic acid bacteria (e.g., lactobacilli, streptococci or lactococci) may be protective against the development and progression of colorectal cancer through several mechanisms such as antioxidant activity, immunomodulation, promoting programmed cell death, antiproliferative effects, and epigenetic modification of cancer cells.[214]
- The Cancer Genome Atlas[53]
- The Colorectal Cancer Atlas integrating genomic and proteomic data pertaining to colorectal cancer tissues and cell lines have been developed.[215]
See also
[edit]References
[edit]- ^ a b c d "General Information About Colon Cancer". NCI. May 12, 2014. Archived from the original on July 4, 2014. Retrieved June 29, 2014.
- ^ a b c d e f g h i j k l Bosman FT (2014). "Chapter 5.5: Colorectal Cancer". In Stewart BW, Wild CP (eds.). World Cancer Report. the International Agency for Research on Cancer, World Health Organization. pp. 392–402. ISBN 978-92-832-0443-5.
- ^ a b c "Colorectal Cancer Prevention (PDQ®)". National Cancer Institute. February 27, 2014. Archived from the original on July 5, 2014. Retrieved June 29, 2014.
- ^ a b Theodoratou E, Timofeeva M, Li X, Meng X, Ioannidis JP (August 2017). "Nature, Nurture, and Cancer Risks: Genetic and Nutritional Contributions to Cancer". Annual Review of Nutrition (Review). 37: 293–320. doi:10.1146/annurev-nutr-071715-051004. PMC 6143166. PMID 28826375.
- ^ a b c d e f g "Colon Cancer Treatment (PDQ®)". NCI. May 12, 2014. Archived from the original on July 5, 2014. Retrieved June 29, 2014.
- ^ a b "SEER Stat Fact Sheets: Colon and Rectum Cancer". NCI. Archived from the original on June 24, 2014. Retrieved June 18, 2014.
- ^ Vos T, Barber RM, Bell B, Bertozzi-Villa A, Biryukov S, Bolliger I, et al. (GBD 2015 Disease and Injury Incidence and Prevalence Collaborators) (October 2016). "Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015". Lancet. 388 (10053): 1545–1602. doi:10.1016/S0140-6736(16)31678-6. PMC 5055577. PMID 27733282.
- ^ a b Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (November 2018). "Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries". CA: A Cancer Journal for Clinicians. 68 (6): 394–424. doi:10.3322/caac.21492. PMID 30207593. S2CID 52188256.
- ^ "Colorectal Cancer Signs and Symptoms | Signs of Colorectal Cancer". www.cancer.org. Retrieved February 8, 2023.
- ^ a b c d e f g Bibbins-Domingo K, Grossman DC, Curry SJ, Davidson KW, Epling JW, García FA, et al. (June 2016). "Screening for Colorectal Cancer: US Preventive Services Task Force Recommendation Statement". JAMA. 315 (23): 2564–2575. doi:10.1001/jama.2016.5989. PMID 27304597.
- ^ "First Colonoscopies Now Recommended at Age 45". ThedaCare. Retrieved December 30, 2022.
- ^ Thorat MA, Cuzick J (December 2013). "Role of aspirin in cancer prevention". Current Oncology Reports. 15 (6): 533–540. doi:10.1007/s11912-013-0351-3. PMID 24114189. S2CID 40187047.
- ^ "Routine aspirin or nonsteroidal anti-inflammatory drugs for the primary prevention of colorectal cancer: recommendation statement". American Family Physician. 76 (1): 109–113. July 2007. PMID 17668849. Archived from the original on July 14, 2014.
- ^ a b Forman D, Ferlay J (2014). "Chapter 1.1: The global and regional burden of cancer". In Stewart BW, Wild CP (eds.). World Cancer Report. the International Agency for Research on Cancer, World Health Organization. pp. 16–53. ISBN 978-92-832-0443-5.
- ^ Alpers DH, Kalloo AN, Kaplowitz N, Owyang C, Powell DW (2008). Yamada T (ed.). Principles of clinical gastroenterology. Chichester, West Sussex: Wiley-Blackwell. p. 381. ISBN 978-1-4051-6910-3. Archived from the original on September 28, 2015.
- ^ Juul JS, Hornung N, Andersen B, Laurberg S, Olesen F, Vedsted P (August 2018). "The value of using the faecal immunochemical test in general practice on patients presenting with non-alarm symptoms of colorectal cancer". British Journal of Cancer. 119 (4): 471–479. doi:10.1038/s41416-018-0178-7. PMC 6133998. PMID 30065255.
- ^ a b Astin M, Griffin T, Neal RD, Rose P, Hamilton W (May 2011). "The diagnostic value of symptoms for colorectal cancer in primary care: a systematic review". The British Journal of General Practice. 61 (586): e231–e243. doi:10.3399/bjgp11X572427. PMC 3080228. PMID 21619747.
- ^ Adelstein BA, Macaskill P, Chan SF, Katelaris PH, Irwig L (May 2011). "Most bowel cancer symptoms do not indicate colorectal cancer and polyps: a systematic review". BMC Gastroenterology. 11: 65. doi:10.1186/1471-230X-11-65. PMC 3120795. PMID 21624112.
- ^ a b Watson AJ, Collins PD (2011). "Colon cancer: a civilization disorder". Digestive Diseases. 29 (2): 222–228. doi:10.1159/000323926. PMID 21734388. S2CID 7640363.
- ^ a b c d e f g h i j k l m n o p q r s t Cunningham D, Atkin W, Lenz HJ, Lynch HT, Minsky B, Nordlinger B, et al. (March 2010). "Colorectal cancer". Lancet. 375 (9719): 1030–1047. doi:10.1016/S0140-6736(10)60353-4. PMID 20304247. S2CID 25299272.
- ^ a b "Colorectal Cancer 2011 Report: Food, Nutrition, Physical Activity, and the Prevention of Colorectal Cancer" (PDF). World Cancer Research Fund & American Institute for Cancer Research. 2011. Archived (PDF) from the original on September 9, 2016.
- ^ Vyas DA, Eisenstein LG, Jones DS (August 27, 2020). Malina D (ed.). "Hidden in Plain Sight — Reconsidering the Use of Race Correction in Clinical Algorithms". New England Journal of Medicine. 383 (9): 874–882. doi:10.1056/NEJMms2004740. ISSN 0028-4793. PMID 32853499. S2CID 221359557.
- ^ Bowles TL, Hu CY, You NY, Skibber JM, Rodriguez-Bigas MA, Chang GJ (May 2013). "An Individualized Conditional Survival Calculator for Patients with Rectal Cancer". Diseases of the Colon & Rectum. 56 (5): 551–559. doi:10.1097/DCR.0b013e31827bd287. ISSN 0012-3706. PMC 3673550. PMID 23575393.
- ^ Lee IM, Shiroma EJ, Lobelo F, Puska P, Blair SN, Katzmarzyk PT (July 2012). "Effect of physical inactivity on major non-communicable diseases worldwide: an analysis of burden of disease and life expectancy". Lancet. 380 (9838): 219–229. doi:10.1016/S0140-6736(12)61031-9. PMC 3645500. PMID 22818936.
- ^ Fedirko V, Tramacere I, Bagnardi V, Rota M, Scotti L, Islami F, et al. (September 2011). "Alcohol drinking and colorectal cancer risk: an overall and dose-response meta-analysis of published studies". Annals of Oncology. 22 (9): 1958–1972. doi:10.1093/annonc/mdq653. PMID 21307158.
- ^ Valtin H (November 2002). ""Drink at least eight glasses of water a day." Really? Is there scientific evidence for "8 x 8"?". American Journal of Physiology. Regulatory, Integrative and Comparative Physiology. 283 (5): R993–1004. doi:10.1152/ajpregu.00365.2002. PMID 12376390. S2CID 2256436.
- ^ Boleij A, van Gelder MM, Swinkels DW, Tjalsma H (November 2011). "Clinical Importance of Streptococcus gallolyticus infection among colorectal cancer patients: systematic review and meta-analysis". Clinical Infectious Diseases. 53 (9): 870–878. doi:10.1093/cid/cir609. PMID 21960713.
- ^ Jans C, Meile L, Lacroix C, Stevens MJ (July 2015). "Genomics, evolution, and molecular epidemiology of the Streptococcus bovis/Streptococcus equinus complex (SBSEC)". Infection, Genetics and Evolution. 33: 419–436. Bibcode:2015InfGE..33..419J. doi:10.1016/j.meegid.2014.09.017. PMID 25233845.
- ^ a b c Abdulamir AS, Hafidh RR, Abu Bakar F (January 2011). "The association of Streptococcus bovis/gallolyticus with colorectal tumors: the nature and the underlying mechanisms of its etiological role". Journal of Experimental & Clinical Cancer Research. 30 (1): 11. doi:10.1186/1756-9966-30-11. PMC 3032743. PMID 21247505.
 This article incorporates text by Ahmed S Abdulamir, Rand R Hafidh, and Fatimah Abu Bakar available under the CC BY 2.0 license.
This article incorporates text by Ahmed S Abdulamir, Rand R Hafidh, and Fatimah Abu Bakar available under the CC BY 2.0 license.
- ^ Arthur JC (June 2020). "Microbiota and colorectal cancer: colibactin makes its mark". Nature Reviews. Gastroenterology & Hepatology. 17 (6): 317–318. doi:10.1038/s41575-020-0303-y. PMID 32317778. S2CID 216033220.
- ^ Jawad N, Direkze N, Leedham SJ (2011). "Inflammatory Bowel Disease and Colon Cancer". Inflammation and Gastrointestinal Cancers. Recent Results in Cancer Research. Vol. 185. pp. 99–115. doi:10.1007/978-3-642-03503-6_6. ISBN 978-3-642-03502-9. PMID 21822822.
- ^ Hu T, Li LF, Shen J, Zhang L, Cho CH (2015). "Chronic inflammation and colorectal cancer: the role of vascular endothelial growth factor". Current Pharmaceutical Design. 21 (21): 2960–2967. doi:10.2174/1381612821666150514104244. PMID 26004415.
- ^ a b c d Triantafillidis JK, Nasioulas G, Kosmidis PA (July 2009). "Colorectal cancer and inflammatory bowel disease: epidemiology, risk factors, mechanisms of carcinogenesis and prevention strategies". Anticancer Research. 29 (7): 2727–2737. PMID 19596953.
- ^ a b Bye WA, Nguyen TM, Parker CE, Jairath V, East JE (September 2017). "Strategies for detecting colon cancer in patients with inflammatory bowel disease". The Cochrane Database of Systematic Reviews. 2017 (9): CD000279. doi:10.1002/14651858.cd000279.pub4. PMC 6483622. PMID 28922695.
- ^ Juhn E, Khachemoune A (2010). "Gardner syndrome: skin manifestations, differential diagnosis and management". American Journal of Clinical Dermatology. 11 (2): 117–122. doi:10.2165/11311180-000000000-00000. PMID 20141232. S2CID 36836169.
- ^ Half E, Bercovich D, Rozen P (October 2009). "Familial adenomatous polyposis". Orphanet Journal of Rare Diseases. 4: 22. doi:10.1186/1750-1172-4-22. PMC 2772987. PMID 19822006.
- ^ Möslein G, Pistorius S, Saeger HD, Schackert HK (March 2003). "Preventive surgery for colon cancer in familial adenomatous polyposis and hereditary nonpolyposis colorectal cancer syndrome". Langenbeck's Archives of Surgery. 388 (1): 9–16. doi:10.1007/s00423-003-0364-8. PMID 12690475. S2CID 21385340.
- ^ Mankaney G, Rouphael C, Burke CA (April 2020). "Serrated Polyposis Syndrome". Clinical Gastroenterology and Hepatology. 18 (4): 777–779. doi:10.1016/j.cgh.2019.09.006. PMID 31520728.
- ^ Fan C, Younis A, Bookhout CE, Crockett SD (March 2018). "Management of Serrated Polyps of the Colon". Current Treatment Options in Gastroenterology. 16 (1): 182–202. doi:10.1007/s11938-018-0176-0. PMC 6284520. PMID 29445907.
- ^ Bourdais R, Rousseau B, Pujals A, Boussion H, Joly C, Guillemin A, et al. (May 2017). "Polymerase proofreading domain mutations: New opportunities for immunotherapy in hypermutated colorectal cancer beyond MMR deficiency". Critical Reviews in Oncology/Hematology. 113: 242–248. doi:10.1016/j.critrevonc.2017.03.027. PMID 28427513.
- ^ Stein U, Walther W, Arlt F, Schwabe H, Smith J, Fichtner I, et al. (January 2009). "MACC1, a newly identified key regulator of HGF-MET signaling, predicts colon cancer metastasis". Nature Medicine. 15 (1): 59–67. doi:10.1038/nm.1889. PMID 19098908. S2CID 8854895.
- ^ Stein U (2013) MACC1 – a novel target for solid cancers. Expert Opin Ther Targets
- ^ Schuebel KE, Chen W, Cope L, Glöckner SC, Suzuki H, Yi JM, et al. (September 2007). "Comparing the DNA hypermethylome with gene mutations in human colorectal cancer". PLOS Genetics. 3 (9): 1709–1723. doi:10.1371/journal.pgen.0030157. PMC 1988850. PMID 17892325.
- ^ "What is the relationship between Ashkenazi Jews and colorectal cancer?". WebMD. Retrieved October 17, 2019.
- ^ a b c Tabibzadeh A, Tameshkel FS, Moradi Y, Soltani S, Moradi-Lakeh M, Ashrafi GH, et al. (October 30, 2020). "Signal transduction pathway mutations in gastrointestinal (GI) cancers: a systematic review and meta-analysis". Scientific Reports. 10 (1): 18713. Bibcode:2020NatSR..1018713T. doi:10.1038/s41598-020-73770-1. ISSN 2045-2322. PMC 7599243. PMID 33127962.
- ^ Ionov Y, Peinado MA, Malkhosyan S, Shibata D, Perucho M (June 1993). "Ubiquitous somatic mutations in simple repeated sequences reveal a new mechanism for colonic carcinogenesis". Nature. 363 (6429): 558–561. Bibcode:1993Natur.363..558I. doi:10.1038/363558a0. PMID 8505985. S2CID 4254940.
- ^ Chakravarthi S, Krishnan B, Madhavan M (1999). "Apoptosis and expression of p53 in colorectal neoplasms". Indian J. Med. Res. 86 (7): 95–102.
- ^ Abdul Khalek FJ, Gallicano GI, Mishra L (November 2010). "Colon cancer stem cells". Gastrointestinal Cancer Research (Suppl 1): S16–S23. PMC 3047031. PMID 21472043.
- ^ a b c d Markowitz SD, Bertagnolli MM (December 2009). "Molecular origins of cancer: Molecular basis of colorectal cancer". The New England Journal of Medicine. 361 (25): 2449–2460. doi:10.1056/NEJMra0804588. PMC 2843693. PMID 20018966.
- ^ Mehlen P, Fearon ER (August 2004). "Role of the dependence receptor DCC in colorectal cancer pathogenesis". Journal of Clinical Oncology. 22 (16): 3420–3428. doi:10.1200/JCO.2004.02.019. PMID 15310786.
- ^ a b c Uhlen M, Zhang C, Lee S, Sjöstedt E, Fagerberg L, Bidkhori G, et al. (August 2017). "A pathology atlas of the human cancer transcriptome". Science. 357 (6352): eaan2507. doi:10.1126/science.aan2507. PMID 28818916.
- ^ Vogelstein B, Kinzler KW (August 2004). "Cancer genes and the pathways they control". Nature Medicine. 10 (8): 789–799. doi:10.1038/nm1087. PMID 15286780. S2CID 205383514.
- ^ a b c Muzny DM, Bainbridge MN, Chang K, Dinh HH, Drummond JA, Fowler G, et al. (Cancer Genome Atlas Network) (July 2012). "Comprehensive molecular characterization of human colon and rectal cancer". Nature. 487 (7407): 330–337. Bibcode:2012Natur.487..330T. doi:10.1038/nature11252. PMC 3401966. PMID 22810696.
- ^ Gatalica Z, Vranic S, Xiu J, Swensen J, Reddy S (July 2016). "High microsatellite instability (MSI-H) colorectal carcinoma: a brief review of predictive biomarkers in the era of personalized medicine". Familial Cancer. 15 (3): 405–412. doi:10.1007/s10689-016-9884-6. PMC 4901118. PMID 26875156.
- ^ a b c Ryan E, Sheahan K, Creavin B, Mohan HM, Winter DC (August 2017). "The current value of determining the mismatch repair status of colorectal cancer: A rationale for routine testing". Critical Reviews in Oncology/Hematology. 116: 38–57. doi:10.1016/j.critrevonc.2017.05.006. PMID 28693799.
- ^ Hissong E, Crowe EP, Yantiss RK, Chen YT (November 2018). "Assessing colorectal cancer mismatch repair status in the modern era: a survey of current practices and re-evaluation of the role of microsatellite instability testing". Modern Pathology. 31 (11): 1756–1766. doi:10.1038/s41379-018-0094-7. PMID 29955148.
- ^ a b c Grady WM, Markowitz SD (March 2015). "The molecular pathogenesis of colorectal cancer and its potential application to colorectal cancer screening". Digestive Diseases and Sciences. 60 (3): 762–772. doi:10.1007/s10620-014-3444-4. PMC 4779895. PMID 25492499.
- ^ Leslie A, Carey FA, Pratt NR, Steele RJ (July 2002). "The colorectal adenoma-carcinoma sequence". The British Journal of Surgery. 89 (7): 845–860. doi:10.1046/j.1365-2168.2002.02120.x. ISSN 0007-1323. PMID 12081733. S2CID 36456541.
- ^ Nguyen LH, Goel A, Chung DC (January 2020). "Pathways of Colorectal Carcinogenesis". Gastroenterology. 158 (2): 291–302. doi:10.1053/j.gastro.2019.08.059. ISSN 0016-5085. PMC 6981255. PMID 31622622.
- ^ Slaughter DP, Southwick HW, Smejkal W (September 1953). "Field cancerization in oral stratified squamous epithelium; clinical implications of multicentric origin". Cancer. 6 (5): 963–968. doi:10.1002/1097-0142(195309)6:5<963::AID-CNCR2820060515>3.0.CO;2-Q. PMID 13094644. S2CID 6736946.
- ^ Giovannucci E, Ogino S (September 2005). "DNA methylation, field effects, and colorectal cancer". Journal of the National Cancer Institute. 97 (18): 1317–1319. doi:10.1093/jnci/dji305. PMID 16174847.
- ^ Bernstein C, Bernstein H, Payne CM, Dvorak K, Garewal H (February 2008). "Field defects in progression to gastrointestinal tract cancers". Cancer Letters. 260 (1–2): 1–10. doi:10.1016/j.canlet.2007.11.027. PMC 2744582. PMID 18164807.
- ^ Nguyen H, Loustaunau C, Facista A, Ramsey L, Hassounah N, Taylor H, et al. (July 2010). "Deficient Pms2, ERCC1, Ku86, CcOI in field defects during progression to colon cancer". Journal of Visualized Experiments (41): 1931. doi:10.3791/1931. PMC 3149991. PMID 20689513. 28 minute video
- ^ Rubin H (March 2011). "Fields and field cancerization: the preneoplastic origins of cancer: asymptomatic hyperplastic fields are precursors of neoplasia, and their progression to tumors can be tracked by saturation density in culture". BioEssays. 33 (3): 224–231. doi:10.1002/bies.201000067. PMID 21254148. S2CID 44981539.
- ^ Tsao JL, Yatabe Y, Salovaara R, Järvinen HJ, Mecklin JP, Aaltonen LA, et al. (February 2000). "Genetic reconstruction of individual colorectal tumor histories". Proceedings of the National Academy of Sciences of the United States of America. 97 (3): 1236–1241. Bibcode:2000PNAS...97.1236T. doi:10.1073/pnas.97.3.1236. PMC 15581. PMID 10655514.
- ^ a b Vogelstein B, Papadopoulos N, Velculescu VE, Zhou S, Diaz LA, Kinzler KW (March 2013). "Cancer genome landscapes". Science. 339 (6127): 1546–1558. Bibcode:2013Sci...339.1546V. doi:10.1126/science.1235122. PMC 3749880. PMID 23539594.
- ^ a b Bernstein C, Nfonsam V, Prasad AR, Bernstein H (March 2013). "Epigenetic field defects in progression to cancer". World Journal of Gastrointestinal Oncology. 5 (3): 43–49. doi:10.4251/wjgo.v5.i3.43. PMC 3648662. PMID 23671730.
- ^ Lochhead P, Chan AT, Nishihara R, Fuchs CS, Beck AH, Giovannucci E, et al. (January 2015). "Etiologic field effect: reappraisal of the field effect concept in cancer predisposition and progression". Modern Pathology. 28 (1): 14–29. doi:10.1038/modpathol.2014.81. PMC 4265316. PMID 24925058.
- ^ Wilbur B, ed. (2009). The World of the Cell (7th ed.). San Francisco, C.
{{cite book}}: CS1 maint: location missing publisher (link) - ^ Kimball's Biology Pages. Archived December 31, 2017, at the Wayback Machine "Oncogenes" Free full text
- ^ Kanwal R, Gupta S (April 2012). "Epigenetic modifications in cancer". Clinical Genetics. 81 (4): 303–311. doi:10.1111/j.1399-0004.2011.01809.x. PMC 3590802. PMID 22082348.
- ^ a b Schnekenburger M, Diederich M (March 2012). "Epigenetics Offer New Horizons for Colorectal Cancer Prevention". Current Colorectal Cancer Reports. 8 (1): 66–81. doi:10.1007/s11888-011-0116-z. PMC 3277709. PMID 22389639.
- ^ a b c d Lao VV, Grady WM (October 2011). "Epigenetics and colorectal cancer". Nature Reviews. Gastroenterology & Hepatology. 8 (12): 686–700. doi:10.1038/nrgastro.2011.173. PMC 3391545. PMID 22009203.
- ^ Klutstein M, Nejman D, Greenfield R, Cedar H (June 2016). "DNA Methylation in Cancer and Aging". Cancer Research. 76 (12): 3446–3450. doi:10.1158/0008-5472.CAN-15-3278. PMID 27256564.
- ^ Jacinto FV, Esteller M (July 2007). "Mutator pathways unleashed by epigenetic silencing in human cancer". Mutagenesis. 22 (4): 247–253. doi:10.1093/mutage/gem009. PMID 17412712.
- ^ Lahtz C, Pfeifer GP (February 2011). "Epigenetic changes of DNA repair genes in cancer". Journal of Molecular Cell Biology. 3 (1): 51–58. doi:10.1093/jmcb/mjq053. PMC 3030973. PMID 21278452.
- ^ "Colorectal Cancer". The Lecturio Medical Concept Library. Retrieved July 22, 2021.
- ^ Coppedè F, Lopomo A, Spisni R, Migliore L (January 2014). "Genetic and epigenetic biomarkers for diagnosis, prognosis and treatment of colorectal cancer". World Journal of Gastroenterology. 20 (4): 943–956. doi:10.3748/wjg.v20.i4.943. PMC 3921546. PMID 24574767.
- ^ Guinney J, Dienstmann R, Wang X, de Reyniès A, Schlicker A, Soneson C, et al. (November 2015). "The consensus molecular subtypes of colorectal cancer". Nature Medicine. 21 (11): 1350–1356. doi:10.1038/nm.3967. PMC 4636487. PMID 26457759.
- ^ Orouji E, Raman AT, Singh AK, Sorokin A, Arslan E, Ghosh AK, et al. (May 2021). "Chromatin state dynamics confers specific therapeutic strategies in enhancer subtypes of colorectal cancer". Gut. 71 (5): 938–949. doi:10.1136/gutjnl-2020-322835. PMC 8745382. PMID 34059508. S2CID 235269540.
- ^ a b "Colorectal Cancer". The Lecturio Medical Concept Library. Retrieved July 10, 2021.
- ^ Awiwi MO, Kaur H, Ernst R, Rauch GM, Morani AC, Stanietzky N, et al. (2023). "Restaging MRI of Rectal Adenocarcinoma after Neoadjuvant Chemoradiotherapy: Imaging Findings and Potential Pitfalls". Radiographics. 43 (4): e220135. doi:10.1148/rg.220135. PMID 36927125. S2CID 257583845.
- ^ Awiwi MO, Kaur H, Ernst R, Rauch GM, Morani AC, Stanietzky N, et al. (April 2023). "Restaging MRI of Rectal Adenocarcinoma after Neoadjuvant Chemoradiotherapy: Imaging Findings and Potential Pitfalls". Radiographics. 43 (4): e220135. doi:10.1148/rg.220135. PMID 36927125. S2CID 257583845.
- ^ Kang H, O'Connell JB, Leonardi MJ, Maggard MA, McGory ML, Ko CY (February 2007). "Rare tumors of the colon and rectum: a national review". International Journal of Colorectal Disease. 22 (2): 183–189. doi:10.1007/s00384-006-0145-2. PMID 16845516. S2CID 34693873.
- ^ "Colon, Rectosigmoid, and Rectum Equivalent Terms and Definitions C180-C189, C199, C209, (Excludes lymphoma and leukemia M9590 – M9992 and Kaposi sarcoma M9140) – Colon Solid Tumor Rules 2018. July 2019 Update" (PDF). National Cancer Institute. Archived (PDF) from the original on January 16, 2020.
- ^ "Colorectal cancer types". Cancer Treatment Centers of America. October 4, 2018. Retrieved January 16, 2020.
- ^ Di Como JA, Mahendraraj K, Lau CS, Chamberlain RS (October 2015). "Adenosquamous carcinoma of the colon and rectum: a population based clinical outcomes study involving 578 patients from the Surveillance Epidemiology and End Result (SEER) database (1973–2010)". Journal of the American College of Surgeons. 221 (4): 56. doi:10.1016/j.jamcollsurg.2015.08.044.
- ^ Whiteside G, Munglani R (September 1998). "TUNEL, Hoechst and immunohistochemistry triple-labelling: an improved method for detection of apoptosis in tissue sections--an update". Brain Research. Brain Research Protocols. 3 (1): 52–53. doi:10.1016/s1385-299x(98)00020-8. PMID 9767106.
- ^ "TNM staging of colorectal carcinoma (AJCC 8th edition)". www.pathologyoutlines.com. Retrieved February 24, 2019.
- ^ Parkin DM, Boyd L, Walker LC (December 2011). "16. The fraction of cancer attributable to lifestyle and environmental factors in the UK in 2010". British Journal of Cancer. 105 (S2): S77–S81. doi:10.1038/bjc.2011.489. PMC 3252065. PMID 22158327.
- ^ Searke D (2006). Cancer Epidemiology and Prevention (3 ed.). Oxford University Press. p. 809. ISBN 978-0-19-974797-9. Archived from the original on September 28, 2015.
- ^ Rennert G (2007). Cancer Prevention. Springer. p. 179. ISBN 978-3-540-37696-5. Archived from the original on October 3, 2015.
- ^ a b "Colorectal Cancer Prevention Overview". National Cancer Institute. March 1, 2018. Retrieved October 26, 2018.
- ^ "Cancer prevention". World Health Organization. Retrieved October 27, 2018.
- ^ Chaplin A, Rodriguez RM, Segura-Sampedro JJ, Ochogavía-Seguí A, Romaguera D, Barceló-Coblijn G (October 2022). "Insights behind the Relationship between Colorectal Cancer and Obesity: Is Visceral Adipose Tissue the Missing Link?". International Journal of Molecular Sciences. 23 (21): 13128. doi:10.3390/ijms232113128. PMC 9655590. PMID 36361914.
- ^ Lauby-Secretan B, Scoccianti C, Loomis D, Grosse Y, Bianchini F, Straif K (August 2016). "Body Fatness and Cancer--Viewpoint of the IARC Working Group". The New England Journal of Medicine. 375 (8): 794–798. doi:10.1056/nejmsr1606602. PMC 6754861. PMID 27557308.
- ^ "Colorectal Cancer – Risk Factors and Prevention". June 25, 2012.
- ^ Willett WC (2014). "Diet, nutrition, and cancer: where next for public health?". In Stewart BW, Wild CP (eds.). World Cancer Report. the International Agency for Research on Cancer, World Health Organization. pp. 432–435. ISBN 978-92-832-0443-5.
- ^ a b Willett WC, Key T, Romieu I (2014). "Chapter 2.6: Diet, obesity, and physical activity". In Stewart BW, Wild CP (eds.). World Cancer Report. the International Agency for Research on Cancer, World Health Organization. pp. 124–133. ISBN 978-92-832-0443-5.
Several large prospective cohort studies of dietary fibre and colon cancer risk have not supported an association, although an inverse relation was seen in the large European Prospective Investigation into Cancer and Nutrition (EPIC) study and a recent meta-analysis. The variation in findings from prospective studies needs to be better understood; dietary fibre is complex and heterogeneous, and the relation with colorectal cancer could differ by dietary source. (p. 127)
- ^ "Colorectal Cancer Prevention Description of Evidence". National Cancer Institute. March 1, 2018. Retrieved October 26, 2018.
- ^ Tuan J, Chen YX (December 18, 2015). "Dietary and Lifestyle Factors Associated with Colorectal Cancer Risk and Interactions with Microbiota: Fiber, Red or Processed Meat and Alcoholic Drinks". Gastrointestinal Tumors. 3 (1). S. Karger AG: 17–24. doi:10.1159/000442831. ISSN 2296-3774. PMC 5040877. PMID 27722153.
- ^ Reynolds A, Mann J, Cummings J, Winter N, Mete E, Te Morenga L (February 2019). "Carbohydrate quality and human health: a series of systematic reviews and meta-analyses". Lancet. 393 (10170): 434–445. doi:10.1016/S0140-6736(18)31809-9. PMID 30638909. S2CID 58632705.
- ^ Song M, Chan AT (January 2019). "Environmental Factors, Gut Microbiota, and Colorectal Cancer Prevention". Clinical Gastroenterology and Hepatology. 17 (2): 275–289. doi:10.1016/j.cgh.2018.07.012. PMC 6314893. PMID 30031175.
Despite the longstanding hypothesis that a high-fiber diet may protect against colorectal cancer... epidemiologic studies associating dietary fiber intake with subsequent risk of colorectal cancer have yielded inconsistent results... Nonetheless, based on existing evidence, the most recent expert report from the World Cancer Research Fund and American Institute for Cancer Research in 2017 concludes that there is probable evidence
- ^ Jabbari M, Pourmoradian S, Eini-Zinab H, Mosharkesh E, Hosseini Balam F, Yaghmaei Y, et al. (November 2022). "Levels of evidence for the association between different food groups/items consumption and the risk of various cancer sites: an umbrella review". International Journal of Food Sciences and Nutrition. 73 (7): 861–874. doi:10.1080/09637486.2022.2103523. PMID 35920747. S2CID 251280745.
- ^ Pérez-Cueto FJ, Verbeke W (April 2012). "Consumer implications of the WCRF's permanent update on colorectal cancer". Meat Science. 90 (4): 977–978. doi:10.1016/j.meatsci.2011.11.032. PMID 22196090.
- ^ Harriss DJ, Atkinson G, Batterham A, George K, Cable NT, Reilly T, et al. (September 2009). "Lifestyle factors and colorectal cancer risk (2): a systematic review and meta-analysis of associations with leisure-time physical activity". Colorectal Disease. 11 (7): 689–701. doi:10.1111/j.1463-1318.2009.01767.x. PMID 19207713. S2CID 8026021.
- ^ Robsahm TE, Aagnes B, Hjartåker A, Langseth H, Bray FI, Larsen IK (November 2013). "Body mass index, physical activity, and colorectal cancer by anatomical subsites: a systematic review and meta-analysis of cohort studies". European Journal of Cancer Prevention. 22 (6): 492–505. doi:10.1097/CEJ.0b013e328360f434. PMID 23591454. S2CID 24764995.
- ^ Kyu HH, Bachman VF, Alexander LT, Mumford JE, Afshin A, Estep K, et al. (August 2016). "Physical activity and risk of breast cancer, colon cancer, diabetes, ischemic heart disease, and ischemic stroke events: systematic review and dose-response meta-analysis for the Global Burden of Disease Study 2013". BMJ. 354: i3857. doi:10.1136/bmj.i3857. PMC 4979358. PMID 27510511.
- ^ Biswas A, Oh PI, Faulkner GE, Bajaj RR, Silver MA, Mitchell MS, et al. (January 2015). "Sedentary time and its association with risk for disease incidence, mortality, and hospitalization in adults: a systematic review and meta-analysis". Annals of Internal Medicine. 162 (2): 123–132. doi:10.7326/M14-1651. PMID 25599350. S2CID 7256176.
- ^ Cooper K, Squires H, Carroll C, Papaioannou D, Booth A, Logan RF, et al. (June 2010). "Chemoprevention of colorectal cancer: systematic review and economic evaluation". Health Technology Assessment. 14 (32): 1–206. doi:10.3310/hta14320. PMID 20594533.
- ^ Emilsson L, Holme Ø, Bretthauer M, Cook NR, Buring JE, Løberg M, et al. (January 2017). "Systematic review with meta-analysis: the comparative effectiveness of aspirin vs. screening for colorectal cancer prevention". Alimentary Pharmacology & Therapeutics. 45 (2): 193–204. doi:10.1111/apt.13857. PMID 27859394.
- ^ Bibbins-Domingo K (June 2016). "Aspirin Use for the Primary Prevention of Cardiovascular Disease and Colorectal Cancer: U.S. Preventive Services Task Force Recommendation Statement". Annals of Internal Medicine. 164 (12): 836–845. doi:10.7326/M16-0577. PMID 27064677.
- ^ Agency for Healthcare Research and Quality. "Aspirin or Nonsteroidal Anti-inflammatory Drugs for the Primary Prevention of Colorectal Cancer". United States Department of Health & Human Services. Archived from the original on January 5, 2016.
2010/2011
- ^ Weingarten MA, Zalmanovici A, Yaphe J (January 2008). "Dietary calcium supplementation for preventing colorectal cancer and adenomatous polyps". The Cochrane Database of Systematic Reviews. 2010 (1): CD003548. doi:10.1002/14651858.CD003548.pub4. PMC 8719254. PMID 18254022.
- ^ Ma Y, Zhang P, Wang F, Yang J, Liu Z, Qin H (October 2011). "Association between vitamin D and risk of colorectal cancer: a systematic review of prospective studies". Journal of Clinical Oncology. 29 (28): 3775–3782. doi:10.1200/JCO.2011.35.7566. PMID 21876081.
- ^ Yin L, Grandi N, Raum E, Haug U, Arndt V, Brenner H (2011). "Meta-analysis: Serum vitamin D and colorectal adenoma risk". Preventive Medicine. 53 (1–2): 10–16. doi:10.1016/j.ypmed.2011.05.013. PMID 21672549.
- ^ "What Can I Do to Reduce My Risk of Colorectal Cancer?". Centers for Disease Control and Prevention. April 2, 2014. Archived from the original on February 26, 2015. Retrieved March 5, 2015.
- ^ a b He J, Efron JE (2011). "Screening for colorectal cancer". Advances in Surgery. 45: 31–44. doi:10.1016/j.yasu.2011.03.006. hdl:2328/11906. PMID 21954677.
- ^ Siegel RL, Ward EM, Jemal A (March 2012). "Trends in colorectal cancer incidence rates in the United States by tumor location and stage, 1992-2008". Cancer Epidemiology, Biomarkers & Prevention. 21 (3): 411–416. doi:10.1158/1055-9965.EPI-11-1020. PMID 22219318.
- ^ Swartz AW, Eberth JM, Josey MJ, Strayer SM (October 2017). "Reanalysis of All-Cause Mortality in the U.S. Preventive Services Task Force 2016 Evidence Report on Colorectal Cancer Screening". Annals of Internal Medicine. 167 (8): 602–603. doi:10.7326/M17-0859. PMC 5823607. PMID 28828493.
- ^ Hewitson P, Glasziou P, Watson E, Towler B, Irwig L (June 2008). "Cochrane systematic review of colorectal cancer screening using the fecal occult blood test (hemoccult): an update". The American Journal of Gastroenterology. 103 (6): 1541–1549. doi:10.1111/j.1572-0241.2008.01875.x. PMID 18479499. S2CID 26338156.
- ^ Lee JK, Liles EG, Bent S, Levin TR, Corley DA (February 2014). "Accuracy of fecal immunochemical tests for colorectal cancer: systematic review and meta-analysis". Annals of Internal Medicine. 160 (3): 171. doi:10.7326/M13-1484. PMC 4189821. PMID 24658694.
- ^ "New pathways could improve bowel cancer screening". NIHR Evidence. September 13, 2021. doi:10.3310/alert_47581. S2CID 239113610. Retrieved August 5, 2022.
- ^ a b Li SJ, Sharples LD, Benton SC, Blyuss O, Mathews C, Sasieni P, et al. (September 2021). "Faecal immunochemical testing in bowel cancer screening: Estimating outcomes for different diagnostic policies". Journal of Medical Screening. 28 (3): 277–285. doi:10.1177/0969141320980501. PMC 8366184. PMID 33342370.
- ^ a b Qaseem A, Crandall CJ, Mustafa RA, Hicks LA, Wilt TJ, Forciea MA, et al. (November 2019). "Screening for Colorectal Cancer in Asymptomatic Average-Risk Adults: A Guidance Statement From the American College of Physicians". Annals of Internal Medicine. 171 (9): 643–654. doi:10.7326/M19-0642. PMC 8152103. PMID 31683290.
- ^ Wolf AM, Fontham ET, Church TR, Flowers CR, Guerra CE, LaMonte SJ, et al. (July 2018). "Colorectal cancer screening for average-risk adults: 2018 guideline update from the American Cancer Society". CA: A Cancer Journal for Clinicians. 68 (4): 250–281. doi:10.3322/caac.21457. PMID 29846947.
- ^ "Screening for Colorectal Cancer". U.S. Preventive Services Task Force. 2008. Archived from the original on February 7, 2015. Retrieved December 19, 2011.
- ^ a b Brenner H, Stock C, Hoffmeister M (April 2014). "Effect of screening sigmoidoscopy and screening colonoscopy on colorectal cancer incidence and mortality: systematic review and meta-analysis of randomised controlled trials and observational studies". BMJ. 348 (apr09 1): g2467. doi:10.1136/bmj.g2467. PMC 3980789. PMID 24922745.
- ^ "Five Things Physicians and Patients Should Question" (PDF). Choosing Wisely: An Initiative of the ABIM Foundation. American Gastroenterological Association. Archived from the original (PDF) on August 9, 2012. Retrieved August 17, 2012.
- ^ Winawer S, Fletcher R, Rex D, Bond J, Burt R, Ferrucci J, et al. (February 2003). "Colorectal cancer screening and surveillance: clinical guidelines and rationale-Update based on new evidence". Gastroenterology. 124 (2): 544–560. doi:10.1053/gast.2003.50044. PMID 12557158. S2CID 29354772.
- ^ Qaseem A, Denberg TD, Hopkins RH, Humphrey LL, Levine J, Sweet DE, et al. (March 2012). "Screening for colorectal cancer: a guidance statement from the American College of Physicians". Annals of Internal Medicine. 156 (5): 378–386. doi:10.7326/0003-4819-156-5-201203060-00010. PMID 22393133.
- ^ Tang V, Boscardin WJ, Stijacic-Cenzer I, Lee SJ (April 2015). "Time to benefit for colorectal cancer screening: survival meta-analysis of flexible sigmoidoscopy trials". BMJ. 350: h1662. doi:10.1136/bmj.h1662. PMC 4399600. PMID 25881903.
- ^ a b Bacchus CM, Dunfield L, Gorber SC, Holmes NM, Birtwhistle R, Dickinson JA, et al. (March 2016). "Recommendations on screening for colorectal cancer in primary care". CMAJ. 188 (5): 340–348. doi:10.1503/cmaj.151125. PMC 4786388. PMID 26903355.
- ^ "NHS Bowel Cancer Screening Programme". cancerscreening.nhs.uk. Archived from the original on November 29, 2014.
- ^ "Home – Bowel Cancer Australia". bowelcanceraustralia.org. Archived from the original on December 24, 2014.
- ^ "Bevolkingsonderzoek darmkanker". rivm.nl. Archived from the original on December 17, 2014.
- ^ Tepus M, Yau TO (July 2020). "Non-Invasive Colorectal Cancer Screening: An Overview". Gastrointestinal Tumors. 7 (3): 62–73. doi:10.1159/000507701. PMC 7445682. PMID 32903904.
- ^ "New pathways could improve bowel cancer screening". NIHR Evidence. September 13, 2021. doi:10.3310/alert_47581. S2CID 239113610.
- ^ Stein A, Atanackovic D, Bokemeyer C (September 2011). "Current standards and new trends in the primary treatment of colorectal cancer". European Journal of Cancer. 47 (Suppl 3): S312–S314. doi:10.1016/S0959-8049(11)70183-6. PMID 21943995.
- ^ Chiorean EG, Nandakumar G, Fadelu T, Temin S, Alarcon-Rozas AE, Bejarano S, et al. (March 2020). "Treatment of Patients With Late-Stage Colorectal Cancer: ASCO Resource-Stratified Guideline". JCO Global Oncology. 6 (6): 414–438. doi:10.1200/JGO.19.00367. PMC 7124947. PMID 32150483.
- ^ Yamashita K, Oka S, Tanaka S, Nagata S, Kuwai T, Furudoi A, et al. (March 1, 2019). "Long-term prognosis after treatment for T1 carcinoma of laterally spreading tumors: a multicenter retrospective study". International Journal of Colorectal Disease. 34 (3): 481–490. doi:10.1007/s00384-018-3203-7. ISSN 1432-1262. PMID 30607579. S2CID 57427824.
- ^ a b McCarthy K, Pearson K, Fulton R, Hewitt J, et al. (Cochrane Colorectal Cancer Group) (December 2012). "Pre-operative chemoradiation for non-metastatic locally advanced rectal cancer". The Cochrane Database of Systematic Reviews. 12: CD008368. doi:10.1002/14651858.CD008368.pub2. PMID 23235660.
- ^ a b "Colorectal (Colon) Cancer". Cleveland Clinic. Retrieved July 9, 2021.
- ^ Böckelman C, Engelmann BE, Kaprio T, Hansen TF, Glimelius B (January 2015). "Risk of recurrence in patients with colon cancer stage II and III: a systematic review and meta-analysis of recent literature". Acta Oncologica. 54 (1): 5–16. doi:10.3109/0284186x.2014.975839. PMID 25430983.
- ^ "Chemotherapy of metastatic colorectal cancer". Prescrire International. 19 (109): 219–224. October 2010. PMID 21180382.
- ^ Fakih MG (June 2015). "Metastatic colorectal cancer: current state and future directions". Journal of Clinical Oncology. 33 (16): 1809–1824. doi:10.1200/JCO.2014.59.7633. PMID 25918280.
- ^ Bendell J (March 14, 2024). "Antiangiogenic Agents in First-Line and Second-Line Therapy for Advanced Colorectal Cancer". Gastrointestinal Cancer Research. 1 (4 Suppl 2). International Society of Gastrointestinal Oncology: S22-8. PMC 2666834. PMID 19365574.
- ^ Shaib W, Mahajan R, El-Rayes B (September 2013). "Markers of resistance to anti-EGFR therapy in colorectal cancer". Journal of Gastrointestinal Oncology. 4 (3): 308–318. doi:10.3978/j.issn.2078-6891.2013.029. PMC 3712296. PMID 23997942.
- ^ Yau TO (October 2019). "Precision treatment in colorectal cancer: Now and the future". JGH Open. 3 (5): 361–369. doi:10.1002/jgh3.12153. PMC 6788378. PMID 31633039.
- ^ Sugarbaker PH, Van der Speeten K (February 2016). "Surgical technology and pharmacology of hyperthermic perioperative chemotherapy". Journal of Gastrointestinal Oncology. 7 (1): 29–44. doi:10.3978/j.issn.2078-6891.2015.105. PMC 4754302. PMID 26941982.
- ^ Segura-Sampedro JJ, Morales-Soriano R (August 2020). "Prophylactic HIPEC with oxaliplatin might be of benefit in T4 and perforated colon cancer: another possible interpretation of the COLOPEC results". Revista Espanola de Enfermedades Digestivas. 112 (8): 666. doi:10.17235/reed.2020.6755/2019. hdl:20.500.13003/12309. PMID 32686435.
- ^ Esquivel J, Sticca R, Sugarbaker P, Levine E, Yan TD, Alexander R, et al. (January 2007). "Cytoreductive surgery and hyperthermic intraperitoneal chemotherapy in the management of peritoneal surface malignancies of colonic origin: a consensus statement. Society of Surgical Oncology". Annals of Surgical Oncology. 14 (1): 128–133. doi:10.1245/s10434-006-9185-7. PMID 17072675. S2CID 21282326.
- ^ Arjona-Sánchez A, Espinosa-Redondo E, Gutiérrez-Calvo A, Segura-Sampedro JJ, Pérez-Viejo E, Concepción-Martín V, et al. (April 2023). "Efficacy and Safety of Intraoperative Hyperthermic Intraperitoneal Chemotherapy for Locally Advanced Colon Cancer: A Phase 3 Randomized Clinical Trial". JAMA Surgery. 158 (7): 683–691. doi:10.1001/jamasurg.2023.0662. PMC 10134040. PMID 37099280.
- ^ a b Troester A, Parikh R, Southwell B, Ester E, Sultan S, Greeno E, et al. (August 20, 2024). "Treatment of stage I-III squamous cell anal cancer: a comparative effectiveness systematic review". JNCI: Journal of the National Cancer Institute. doi:10.1093/jnci/djae195. ISSN 0027-8874.
- ^ DeVita VT, Lawrence TS, Rosenberg SA (2008). DeVita, Hellman, and Rosenberg's Cancer: Principles & Practice of Oncology. Lippincott Williams & Wilkins. pp. 1258–. ISBN 978-0-7817-7207-5.
- ^ Huh JW, Tanksley J, Chino J, Willett CG, Dewhirst MW (July 1, 2020). "Long-term Consequences of Pelvic Irradiation: Toxicities, Challenges, and Therapeutic Opportunities with Pharmacologic Mitigators". Clinical Cancer Research. 26 (13): 3079–3090. doi:10.1158/1078-0432.CCR-19-2744. ISSN 1078-0432. Retrieved October 24, 2024.
- ^ a b Feeney G, Sehgal R, Sheehan M, Hogan A, Regan M, Joyce M, et al. (September 2019). "Neoadjuvant radiotherapy for rectal cancer management". World Journal of Gastroenterology. 25 (33): 4850–4869. doi:10.3748/wjg.v25.i33.4850. PMC 6737323. PMID 31543678.
- ^ Li Y, Wang J, Ma X, Tan L, Yan Y, Xue C, et al. (2016). "A Review of Neoadjuvant Chemoradiotherapy for Locally Advanced Rectal Cancer". International Journal of Biological Sciences. 12 (8): 1022–1031. doi:10.7150/ijbs.15438. PMC 4971740. PMID 27489505.
- ^ Cao C, Wang D, Tian DH, Wilson-Smith A, Huang J, Rimner A (December 2019). "A systematic review and meta-analysis of stereotactic body radiation therapy for colorectal pulmonary metastases". Journal of Thoracic Disease. 11 (12): 5187–5198. doi:10.21037/jtd.2019.12.12. PMC 6988072. PMID 32030236.
- ^ Boland PM, Ma WW (May 2017). "Immunotherapy for Colorectal Cancer". Cancers. 9 (5): 50. doi:10.3390/cancers9050050. PMC 5447960. PMID 28492495.
- ^ a b Syn NL, Teng MW, Mok TS, Soo RA (December 2017). "De-novo and acquired resistance to immune checkpoint targeting". The Lancet. Oncology. 18 (12): e731–e741. doi:10.1016/s1470-2045(17)30607-1. PMID 29208439.
- ^ Borras, DM, Verbandt, S, Ausserhofer, M, et al. (November 2023). "Single cell dynamics of tumor specificity vs bystander activity in CD8+ T cells define the diverse immune landscapes in colorectal cancer". Cell Discovery. 9 (114): 114. doi:10.1038/s41421-023-00605-4. PMC 10652011. PMID 37968259.
- ^ "FDA grants accelerated approval to pembrolizumab for first tissue/site agnostic indication". U.S. Food and Drug Administration. February 9, 2019.
- ^ Cercek A, Lumish M, Sinopoli J, Weiss J, Shia J, Lamendola-Essel M, et al. (June 2022). "PD-1 Blockade in Mismatch Repair–Deficient, Locally Advanced Rectal Cancer". The New England Journal of Medicine. 386 (25): 2363–2376. doi:10.1056/NEJMoa2201445. PMC 9492301. PMID 35660797. S2CID 249395846.
- ^ "Palliative or Supportive Care". American Cancer Society. Archived from the original on August 21, 2014. Retrieved August 20, 2014.
- ^ "ASCO Provisional Clinical Opinion: The Integration of Palliative Care into Standard Oncology Care". ASCO. Archived from the original on August 21, 2014. Retrieved 20 August 2014.
- ^ Higginson IJ, Evans CJ (September–October 2010). "What is the evidence that palliative care teams improve outcomes for cancer patients and their families?". Cancer Journal. 16 (5): 423–435. doi:10.1097/PPO.0b013e3181f684e5. PMID 20890138. S2CID 39881122.
- ^ Wasserberg N, Kaufman HS (December 2007). "Palliation of colorectal cancer". Surgical Oncology. 16 (4): 299–310. doi:10.1016/j.suronc.2007.08.008. PMID 17913495.
- ^ Amersi F, Stamos MJ, Ko CY (July 2004). "Palliative care for colorectal cancer". Surgical Oncology Clinics of North America. 13 (3): 467–477. doi:10.1016/j.soc.2004.03.002. PMID 15236729.
- ^ a b c d Son H, Son YJ, Kim H, Lee Y (June 8, 2018). "Effect of psychosocial interventions on the quality of life of patients with colorectal cancer: a systematic review and meta-analysis". Health and Quality of Life Outcomes. 16 (1): 119. doi:10.1186/s12955-018-0943-6. ISSN 1477-7525. PMC 5994008. PMID 29884182.
- ^ a b c d Peng YN, Huang ML, Kao CH (January 31, 2019). "Prevalence of Depression and Anxiety in Colorectal Cancer Patients: A Literature Review". International Journal of Environmental Research and Public Health. 16 (3): 411. doi:10.3390/ijerph16030411. ISSN 1660-4601. PMC 6388369. PMID 30709020.
- ^ a b Cheng V, Oveisi N, McTaggart-Cowan H, Loree JM, Murphy RA, De Vera MA (2022). "Colorectal Cancer and Onset of Anxiety and Depression: A Systematic Review and Meta-Analysis". Current Oncology. 29 (11): 8751–8766. doi:10.3390/curroncol29110689. ISSN 1718-7729. PMC 9689519. PMID 36421342.
- ^ Mosher CE, Winger JG, Given BA, Helft PR, O'Neil BH (2016). "Mental health outcomes during colorectal cancer survivorship: a review of the literature: Mental health in colorectal cancer survivorship". Psycho-Oncology. 25 (11): 1261–1270. doi:10.1002/pon.3954. PMC 4894828. PMID 26315692.
- ^ a b c Reynolds LM, Consedine NS, Pizarro DA, Bissett IP (2013). "Disgust and Behavioral Avoidance in Colorectal Cancer Screening and Treatment: A Systematic Review and Research Agenda". Cancer Nursing. 36 (2): 122–130. doi:10.1097/NCC.0b013e31826a4b1b. ISSN 0162-220X. PMID 23047793.
- ^ a b c d e f Phelan SM, Griffin JM, Jackson GL, Zafar SY, Hellerstedt W, Stahre M, et al. (2013). "Stigma, perceived blame, self-blame, and depressive symptoms in men with colorectal cancer". Psycho-Oncology. 22 (1): 65–73. doi:10.1002/pon.2048. ISSN 1057-9249. PMC 6000725. PMID 21954081.
- ^ a b Hoon LS, Chi Sally CW, Hong-Gu H (2013). "Effect of psychosocial interventions on outcomes of patients with colorectal cancer: A review of the literature". European Journal of Oncology Nursing. 17 (6): 883–891. doi:10.1016/j.ejon.2013.05.001. ISSN 1462-3889. PMID 23759360.
- ^ Meng X, Wang X, Dong Z (October 1, 2021). "Impact of non-pharmacological interventions on quality of life, anxiety, and depression scores in patients with colorectal cancer: a systematic review and meta-analysis of randomized controlled trials". Supportive Care in Cancer. 29 (10): 5635–5652. doi:10.1007/s00520-021-06185-x. ISSN 1433-7339. PMID 33786669.
- ^ a b c d Hua H, Jiang Q, Sun P, Xu X (May 5, 2023). "Risk factors for early-onset colorectal cancer: systematic review and meta-analysis". Frontiers in Oncology. 13. doi:10.3389/fonc.2023.1132306. ISSN 2234-943X. PMC 10196487. PMID 37213277.
- ^ a b "National Comprehensive Cancer Network" (PDF). nccn.org. Archived (PDF) from the original on March 25, 2009.
- ^ a b Desch CE, Benson AB, Somerfield MR, Flynn PJ, Krause C, Loprinzi CL, et al. (November 2005). "Colorectal cancer surveillance: 2005 update of an American Society of Clinical Oncology practice guideline". Journal of Clinical Oncology. 23 (33): 8512–8519. doi:10.1200/JCO.2005.04.0063. PMID 16260687.
- ^ Jeffery M, Hickey BE, Hider PN (September 2019). "Follow-up strategies for patients treated for non-metastatic colorectal cancer". The Cochrane Database of Systematic Reviews. 2019 (9): CD002200. doi:10.1002/14651858.CD002200.pub4. PMC 6726414. PMID 31483854.
- ^ Betof AS, Dewhirst MW, Jones LW (March 2013). "Effects and potential mechanisms of exercise training on cancer progression: a translational perspective". Brain, Behavior, and Immunity. 30 (Suppl): S75–S87. doi:10.1016/j.bbi.2012.05.001. PMC 3638811. PMID 22610066.
- ^ a b Ballard-Barbash R, Friedenreich CM, Courneya KS, Siddiqi SM, McTiernan A, Alfano CM (June 2012). "Physical activity, biomarkers, and disease outcomes in cancer survivors: a systematic review". Journal of the National Cancer Institute. 104 (11): 815–840. doi:10.1093/jnci/djs207. PMC 3465697. PMID 22570317.
- ^ a b McGettigan M, Cardwell CR, Cantwell MM, Tully MA (May 2020). "Physical activity interventions for disease-related physical and mental health during and following treatment in people with non-advanced colorectal cancer". The Cochrane Database of Systematic Reviews. 2020 (5): CD012864. doi:10.1002/14651858.cd012864.pub2. PMC 7196359. PMID 32361988.
- ^ Strømme JM, Johannessen B, Skotheim RI (2023). "Deviating alternative splicing as a molecular subtype of microsatellite stable colorectal cancer". JCO Clinical Cancer Informatics. 7 (7): e2200159. doi:10.1200/CCI.22.00159. hdl:10852/108838. PMID 36821799.
- ^ Osterman E, Glimelius B (September 2018). "Recurrence Risk After Up-to-Date Colon Cancer Staging, Surgery, and Pathology: Analysis of the Entire Swedish Population". Diseases of the Colon and Rectum. 61 (9): 1016–1025. doi:10.1097/dcr.0000000000001158. PMID 30086050. S2CID 51934598.
- ^ a b Doroudian S, Osterman E, Glimelius B (June 9, 2024). "Risk Factors for Recurrence After Surgery for Rectal Cancer in a Modern, Nationwide Population-Based Cohort". Annals of Surgical Oncology. doi:10.1245/s10434-024-15552-x. ISSN 1068-9265. PMC 11300512. PMID 38853216.
- ^ Nors J, Iversen LH, Erichsen R, Gotschalck KA, Andersen CL (January 1, 2024). "Incidence of Recurrence and Time to Recurrence in Stage I to III Colorectal Cancer: A Nationwide Danish Cohort Study". JAMA Oncology. 10 (1): 54–62. doi:10.1001/jamaoncol.2023.5098. ISSN 2374-2437. PMC 10654928. PMID 37971197.
- ^ Nors J, Gotschalck KA, Erichsen R, Andersen CL (June 1, 2024). "Incidence of late recurrence and second primary cancers 5-10 years after non-metastatic colorectal cancer". International Journal of Cancer. 154 (11): 1890–1899. doi:10.1002/ijc.34871. PMID 38323453.
- ^ Zacharakis M, Xynos ID, Lazaris A, Smaro T, Kosmas C, Dokou A, et al. (February 2010). "Predictors of survival in stage IV metastatic colorectal cancer". Anticancer Research. 30 (2): 653–660. PMID 20332485.
- ^ Agabegi ED, Agabegi SS (2008). Step-Up to Medicine (Step-Up Series). Hagerstwon, MD: Lippincott Williams & Wilkins. ISBN 978-0-7817-7153-5.
- ^ Hong Y (June 30, 2020). "Clinical study of colorectal cancer operation: Survival analysis". Korean Journal of Clinical Oncology 2020. 16 (1): 3–8. doi:10.14216/kjco.20002. PMC 9942716. PMID 36945303.
- ^ "Five-Year Survival Rates". National Cancer Institute. Retrieved May 9, 2021.
- ^ Xu Z, Becerra AZ, Fleming FJ, Aquina CT, Dolan JG, Monson JR, et al. (October 2019). "Treatments for Stage IV Colon Cancer and Overall Survival". The Journal of Surgical Research. 242: 47–54. doi:10.1016/j.jss.2019.04.034. PMID 31071604. S2CID 149443256.
- ^ Drageset S, Lindstrøm TC, Underlid K (April 2016). ""I just have to move on": Women's coping experiences and reflections following their first year after primary breast cancer surgery". European Journal of Oncology Nursing. 21: 205–211. doi:10.1016/j.ejon.2015.10.005. PMID 26521054.
- ^ Restivo A, Zorcolo L, D'Alia G, Cocco F, Cossu A, Scintu F, et al. (February 2016). "Risk of complications and long-term functional alterations after local excision of rectal tumors with transanal endoscopic microsurgery (TEM)". International Journal of Colorectal Disease. 31 (2): 257–266. doi:10.1007/s00384-015-2371-y. PMID 26298182. S2CID 29087556.
- ^ Bregendahl S, Emmertsen KJ, Lindegaard JC, Laurberg S (January 2015). "Urinary and sexual dysfunction in women after resection with and without preoperative radiotherapy for rectal cancer: a population-based cross-sectional study". Colorectal Disease. 17 (1): 26–37. doi:10.1111/codi.12758. PMID 25156386. S2CID 42069306.
- ^ Ramirez M, McMullen C, Grant M, Altschuler A, Hornbrook MC, Krouse RS (December 2009). "Figuring out sex in a reconfigured body: experiences of female colorectal cancer survivors with ostomies". Women & Health. 49 (8): 608–624. doi:10.1080/03630240903496093. PMC 2836795. PMID 20183104.
- ^ Steele N, Haigh R, Knowles G, Mackean M (September 2007). "Carcinoembryonic antigen (CEA) testing in colorectal cancer follow up: what do patients think?". Postgraduate Medical Journal. 83 (983): 612–614. doi:10.1136/pgmj.2007.059634. PMC 2600007. PMID 17823231.
- ^ McGeechan GJ, Byrnes K, Campbell M, Carthy N, Eberhardt J, Paton W, et al. (January 2021). "A systematic review and qualitative synthesis of the experience of living with colorectal cancer as a chronic illness". Psychology & Health. 37 (3): 350–374. doi:10.1080/08870446.2020.1867137. PMID 33499649. S2CID 231771176.
- ^ Lozano R, Naghavi M, Foreman K, Lim S, Shibuya K, Aboyans V, et al. (December 2012). "Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010". Lancet. 380 (9859): 2095–2128. doi:10.1016/S0140-6736(12)61728-0. hdl:10536/DRO/DU:30050819. PMC 10790329. PMID 23245604. S2CID 1541253.
- ^ WHO (February 2010). "Cancer". World Health Organization. Archived from the original on December 29, 2010. Retrieved January 5, 2011.
- ^ Merika E, Saif MW, Katz A, Syrigos K, Syrigos C, Morse M (2010). "Review. Colon cancer vaccines: an update". In Vivo. 24 (5): 607–628. PMID 20952724.
- ^ Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM (2010). "Colorectal Cancer Incidence, Mortality and Prevalence Worldwide in 2008 – Summary". Archived from the original on October 17, 2012.; "GLOBOCAN 2008 v2.0, Cancer Incidence and Mortality Worldwide: IARC CancerBase No. 10". Lyon, France: International Agency for Research on Cancer. Archived from the original on May 8, 2011.
- ^ a b "Colorectal cancer: Statistics". Cancer.net, American Society of Clinical Oncology. February 2022. Retrieved May 13, 2022.
- ^ Katella K. "Colorectal Cancer: What Millennials and Gen Zers Need to Know". YaleMedicine.
- ^ "Bowel cancer | About bowel cancer | Cancer Research UK". www.cancerresearchuk.org. Archived from the original on March 9, 2017. Retrieved May 12, 2017.
- ^ Cancer in Australia: an Overview, 2014. Cancer series No 90. Cat. No. CAN 88. Canberra: Australian Institute of Health and Welfare. 2014. ISBN 978-1-74249-677-1.
- ^ Cancer in Papua New Guinea: an Overview, 2016. Cancer series No. 176. Cat. No. CAN 88. Papua New Guinea Department of Health. 2016.
- ^ a b c d e Puzzono M, Mannucci A, Grannò S, Zuppardo RA, Galli A, Danese S, et al. (2021). "The Role of Diet and Lifestyle in Early-Onset Colorectal Cancer: A Systematic Review". Cancers. 13 (23): 5933. doi:10.3390/cancers13235933. ISSN 2072-6694. PMC 8657307. PMID 34885046.
- ^ a b c d e f Vuik FE, Nieuwenburg SA, Bardou M, Lansdorp-Vogelaar I, Dinis-Ribeiro M, Bento MJ, et al. (2019). "Increasing incidence of colorectal cancer in young adults in Europe over the last 25 years". Gut. 68 (10): 1820–1826. doi:10.1136/gutjnl-2018-317592. ISSN 0017-5749. PMC 6839794. PMID 31097539.
- ^ Unanue-Arza S, Solís-Ibinagagoitia M, Díaz-Seoane M, Mosquera-Metcalfe I, Idigoras I, Bilbao I, et al. (December 12, 2020). "Inequalities and risk factors related to non-participation in colorectal cancer screening programmes: a systematic review". The European Journal of Public Health. 31 (2): 346–355. doi:10.1093/eurpub/ckaa203. ISSN 1101-1262. PMC 8071594. PMID 33313657.
- ^ Rehemtulla A (December 2010). "Dinosaurs and ancient civilizations: reflections on the treatment of cancer". Neoplasia. 12 (12): 957–968. doi:10.1593/neo.101588. PMC 3003131. PMID 21170260.
- ^ Zhong L, Zhang X, Covasa M (June 2014). "Emerging roles of lactic acid bacteria in protection against colorectal cancer". World Journal of Gastroenterology. 20 (24): 7878–7886. doi:10.3748/wjg.v20.i24.7878. PMC 4069315. PMID 24976724.
- ^ "Colorectal Cancer Atlas". Archived from the original on January 13, 2016.