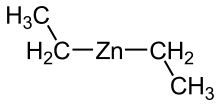
| |

| |
| Names | |
|---|---|
| IUPAC name
diethylzinc
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.008.330 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| UN number | 1366 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| (C2H5)2Zn | |
| Molar mass | 123.50 g/mol |
| Density | 1.205 g/mL |
| Melting point | −28 °C (−18 °F; 245 K) |
| Boiling point | 117 °C (243 °F; 390 K) |
| Reacts | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Flammable and corrosive liquid, pyrophoric in air, may explode in contact with water. |
| GHS labelling: | |
  
| |
| Danger | |
| H225, H250, H260, H302+H312+H332, H314, H410 | |
| P210, P222, P223, P231+P232, P233, P240, P241, P242, P243, P260, P264, P273, P280, P301+P330+P331, P302+P334, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P335+P334, P363, P370+P378, P391, P402+P404, P403+P235, P405, P422, P501 | |
| NFPA 704 (fire diamond) | |
| Safety data sheet (SDS) | External MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Diethylzinc (C2H5)2Zn, or DEZ, is a highly pyrophoric and reactive organozinc compound consisting of a zinc center bound to two ethyl groups. This colourless liquid is an important reagent in organic chemistry. It is available commercially as a solution in hexanes, heptane, or toluene, or as a pure liquid.
Synthesis
[edit]Edward Frankland first reported the compound in 1848 from zinc and ethyl iodide, the first organozinc compound discovered.[2][3] He improved the synthesis by using diethyl mercury as starting material.[4] The contemporary synthesis consists of the reaction of a 1:1 mixture of ethyl iodide and ethyl bromide with a zinc-copper couple, a source of reactive zinc.[5]
Structure
[edit]The compound crystallizes in a tetragonal body-centered unit cell of space group symmetry I41md. In the solid-state diethylzinc shows nearly linear Zn centres. The Zn-C bonds measure 194.8(5) pm, while the C-Zn-C angle is slightly bent with 176.2(4)°.[6] The structure of the gas-phase shows a very similar Zn-C distance (195.0(2) pm).[7]
Uses
[edit]Despite its highly pyrophoric nature, diethylzinc is an important chemical reagent. It is used in organic synthesis as a source of the ethyl carbanion in addition reactions to carbonyl groups. For example, the asymmetric addition of an ethyl group to benzaldehyde[8] and imines.[9] Additionally, it is commonly used in combination with diiodomethane as a Simmons-Smith reagent to convert alkenes into cyclopropyl groups.[10][11] It is less nucleophilic than related alkyllithium and Grignard reagents, so it may be used when a "softer" nucleophile is needed. It is also used extensively in materials science chemistry as a zinc source in the synthesis of nanoparticles. Particularly in the formation of the zinc sulfide shell for core/shell-type quantum dots.[12] While in polymer chemistry, it can be used as part of the catalyst for a chain shuttling polymerization reaction, whereby it participates in living polymerization.[13]
Diethylzinc is not limited to only being used in chemistry. Because of its high reactivity toward air, it was used in small quantities as a hypergolic or "self igniting" liquid rocket fuel[14]: 9 [15]: 323 —it ignites on contact with oxidizer, so the rocket motor need only contain a pump, without a spark source for ignition. Diethylzinc was also investigated by the United States Library of Congress as a potential means of mass deacidification of books printed on wood pulp paper. Diethylzinc vapour would, in theory, neutralize acid residues in the paper, leaving slightly alkaline zinc oxide residues. Although initial results were promising, the project was abandoned. A variety of adverse results prevented the method's adoption. Most infamously, the final prototype suffered damage in a series of diethylzinc explosions from trace amounts of water vapor in the chamber. This led the authors of the study to humorously comment:
It has also been established that tight or loose packing of books; the amount of alkaline reserve; reactions of DEZ with degradation products, unknown paper chemicals and adhesives; phases of the moon and the positions of various planets and constellations do not have any influence on the observed adverse effects of DEZ treatment.[16]
In microelectronics, diethylzinc is used as a doping agent.[citation needed]
For corrosion protection in nuclear reactors of the light water reactor design, depleted zinc oxide is produced by first passing diethylzinc through an enrichment centrifuge.
The pyrophoricity of diethylzinc can be used to test the inert atmosphere inside a glovebox. An oxygen concentration of only a few parts per million will cause a bottle of diethylzinc to fume when opened.[17]
Safety
[edit]Diethylzinc may explode when mixed with water and can spontaneously ignite upon contact with air. It should therefore be handled using air-free techniques.
References
[edit]- ^ "New Environment Inc. - NFPA Chemicals".
- ^ E. Frankland (1850). "On the isolation of the organic radicals". Quarterly Journal of the Chemical Society. 2 (3): 263–296. doi:10.1039/QJ8500200263.
- ^ Dietmar Seyferth (2001). "Zinc Alkyls, Edward Frankland, and the Beginnings of Main-Group Organometallic Chemistry". Organometallics. 20 (14): 2940–2955. doi:10.1021/om010439f.
- ^ E. Frankland, B. F. Duppa (1864). "On a new reaction for the production of the zinc-compounds of the alkyl-radical". Journal of the Chemical Society. 17: 29–36. doi:10.1039/JS8641700029.
- ^ C. R. Noller (1943). "Diethyl Zinc". Organic Syntheses; Collected Volumes, vol. 2, p. 184.
- ^ John Bacsa; Felix Hanke; Sarah Hindley; Rajesh Odedra; George R. Darling; Anthony C. Jones; Alexander Steiner (2011). "The Solid State Structures of Dimethylzinc and Diethylzinc". Angewandte Chemie International Edition. 50 (49): 11685–11687. doi:10.1002/anie.201105099. PMC 3326375. PMID 21919175.
- ^ A. Haaland; J. C. Green; G. S. McGrady; A. J. Downs; E. Gullo; M. J. Lyall; J. Timberlake (2003). "The length, strength and polarity of metal–carbon bonds: dialkylzinc compounds studied by density functional theory calculations, gas electron diffraction and photoelectron spectroscopy". Dalton Transactions (22): 4356–4366. doi:10.1039/B306840B.
- ^ Masato Kitamura, Hiromasa Oka, Seiji Suga, and Ryōji Noyori (2004). "Catalytic Enantioselective Addition of Dialkylzincs to Aldehydes Using (2S)-(−)-3-exo-(Dimethylamino)isoborneol [(2S)-DAIB]: (S)-1-Phenyl-1-propanol". Organic Syntheses
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 10, p. 635. - ^ Jean-Nicolas Desrosiers, Alexandre Côté, Alessandro A. Boezio, and André B. Charette (2005). "Preparation of Enantiomerically Enriched (1S)-1-Phenylpropan-1-amine Hydrochloride by a Catalytic Addition of Diorganozinc Reagents to Imines". Organic Syntheses. 83: 5
{{cite journal}}: CS1 maint: multiple names: authors list (link). - ^ André B. Charette and Hélène Lebel (2004). "(2S,3S)-(+)-(3-Phenylcyclopropyl)methanol". Organic Syntheses; Collected Volumes, vol. 10, p. 613.
- ^ Yoshihiko Ito, Shotaro Fujii, Masashi Nakatuska, Fumio Kawamoto, and Takeo Saegusa (1988). "One-Carbon Ring Expansion of Cycloalkanones to Conjugated Cycloalkenones: 2-Cyclohepten-1-one". Organic Syntheses
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 6, p. 327. - ^ Dmitri V. Talapin; Ivo Mekis; Stephan Götzinger; Andreas Kornowski; Oliver Benson; Horst Weller† (2004). "CdSe/CdS/ZnS and CdSe/ZnSe/ZnS Core−Shell−Shell Nanocrystals". Journal of Physical Chemistry B. 108 (49): 18826–18831. doi:10.1021/jp046481g.
- ^ Mitsuo Sawamoto; Chihiro Okamoto; Toshinobu Higashimura (1987). "Hydrogen iodide/zinc iodide: a new initiating system for living cationic polymerization of vinyl ethers at room temperature". Macromolecules. 20 (11): 2693–2697. Bibcode:1987MaMol..20.2693S. doi:10.1021/ma00177a010.
- ^ Clark, John Drury (23 May 2018). Ignition!: An Informal History of Liquid Rocket Propellants. Rutgers University Press. p. 302. ISBN 978-0-8135-9918-2. OCLC 281664.
- ^ Sutton, George P.; Biblarz, Oscar (2001). Rocket Propulsion Elements - Seventh Edition (PDF). John Wiley & Sons, Inc. ISBN 0-471-32642-9. Archived (PDF) from the original on 28 February 2022.
- ^ Kenneth E. Harris; Chandru J. Shahani (2004), Mass Deacidification: An Initiative To Refine The Diethyl Zinc Process (PDF), Washington, D.C.: Library of Congress, archived from the original (PDF) on 2013-05-14
- ^ Shriver, Duward F.; Drezdzon, Mark A. (1986). The Manipulation of Air-Sensitive Compounds. John Wiley & Sons. p. 57. ISBN 0-471-86773-X.
External links
[edit]- Demonstration of the ignition of diethylzinc in air Video - University of Nottingham
