 | |
| Clinical data | |
|---|---|
| Trade names | Amatine, Gutron, Orvaten, ProAmatine, others |
| Other names | ST-1085; TS-701; 3,6-Dimethoxy-β-hydroxy-N-aminoethanonyl-2-phenylethylamine; 2-Amino-N-[2-(2,5-dimethoxyphenyl)-2-hydroxyethyl]acetamide; 1-2',5'-Dimethoxyphenyl-1)-2 glycinamidoethanol |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a616030 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth[1] |
| Drug class | α1-Adrenergic receptor agonist; Antihypotensive agent; Vasopressor |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 93% (as desglymidodrine)[3][4][1] |
| Metabolism | Deglycination[3][1][4] |
| Metabolites | • Desglymidodrine[3][1][4] |
| Onset of action | ≤1 hour[3] |
| Elimination half-life | Midodrine: 0.5 hours[4] Desglymidodrine: 2–4 hours[4][1] |
| Duration of action | 2–6 hours[3][4] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.151.349 100.050.842, 100.151.349 |
| Chemical and physical data | |
| Formula | C12H18N2O4 |
| Molar mass | 254.286 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
| |
| |
| (verify) | |
Midodrine, sold under the brand names ProAmatine and Orvaten among others, is a vasopressor or antihypotensive medication used to treat orthostatic hypotension (low blood pressure when standing) and urinary incontinence.[3][5][1] It is taken by mouth.[3][1]
Side effects of midodrine include hypertension (high blood pressure), paresthesia, itching, goosebumps, chills, urinary urgency, urinary retention, and urinary frequency.[3] Midodrine is a prodrug of its active metabolite desglymidodrine.[3][1] This metabolite acts as a selective agonist of the α1-adrenergic receptor.[3][1] This in turn results in vasoconstriction and increased blood pressure.[3][1]
Midodrine was discovered by 1971[6] and was introduced for medical use in the United States in 1996.[citation needed]
Medical uses
[edit]Midodrine is indicated for the treatment of symptomatic orthostatic hypotension. It can reduce dizziness and faints by about a third, but can be limited by troublesome goose bumps, skin itch, gastrointestinal discomfort, chills, elevated blood pressure while lying down, and urinary retention.[7] A meta-analysis of clinical trials of midodrine or droxidopa in patients with low blood pressure when standing found that midodrine increased standing blood pressure more than droxidopa but that midodrine but not droxidopa increased the risk of high blood pressure when lying down.[8] Small studies have also shown that midodrine can be used to prevent excessive drops in blood pressure in people requiring dialysis.[9]
Midodrine has been used in the complications of cirrhosis. It is also used with octreotide for hepatorenal syndrome; the proposed mechanism is constriction of splanchnic vessels and dilation of renal vasculature. Studies have not been sufficiently well conducted to show a clear place for midodrine.[10]
Midodrine is used off-label to increase blood pressure in the treatment of postural orthostatic tachycardia syndrome (POTS) where increased transduction of venous alpha 1 adrenergic receptors increases venous return.[11][12][13]
Available forms
[edit]Midodrine is available in the form of 2.5, 5, and 10 mg oral tablets.[3][1]
Contraindications
[edit]Midodrine is contraindicated in patients with severe organic heart disease, acute kidney disease, urinary retention, pheochromocytoma or thyrotoxicosis. Midodrine should not be used in patients with persistent and excessive supine hypertension.[14]
Side effects
[edit]Headache, feeling of pressure or fullness in the head, vasodilation or flushing face, scalp tingling, confusion or thinking abnormality, dry mouth, nervousness or anxiety, and rash, among others.[15]
Pharmacology
[edit]Mechanism of action
[edit]Midodrine is a prodrug which forms the active metabolite, desglymidodrine, which is an α1-adrenergic receptor agonist and exerts its actions via activation of α1-adrenergic receptors of the arteriolar and venous vasculature, producing an increase in vascular tone and elevation of blood pressure. Desglymidodrine does not stimulate cardiac β-adrenergic receptors.

Pharmacokinetics
[edit]After oral administration, midodrine is rapidly absorbed. The plasma levels of the prodrug peak after about half an hour, and decline with a half-life of approximately 25 minutes, while the metabolite reaches peak blood concentrations about 1 to 2 hours after a dose of midodrine and has a half-life of about 3 to 4 hours. The absolute bioavailability of midodrine (measured as desglymidodrine) is 93%.[1]
Midodrine and desglymidodrine diffuse poorly across the blood–brain barrier and are therefore peripherally selective and are not associated with effects in the central nervous system.[16][17][18]
Neither midodrine nor desglymidodrine are substrates of monoamine oxidase.[3]
Chemistry
[edit]Midodrine, also known as 3,6-dimethoxy-β-hydroxy-N-aminoethanonyl-2-phenylethylamine, is a substituted phenethylamine derivative.[4]
Midodrine is an odorless, white, crystalline powder, soluble in water and sparingly soluble in methanol.[19]
Midodrine's experimental log P is -0.5 and its predicted log P ranges from -0.49 to -0.95.[20][1] The predicted log P of its active metabolite desglymidodrine ranges from -0.01 to 0.15.[21][22]
Stereochemistry
[edit]Midodrine contains a stereocenter and consists of two enantiomers, making it a racemate; i.e., a 1:1 mixture of (R)- and (S)-forms:[23]
| Enantiomers of midodrine | |
|---|---|
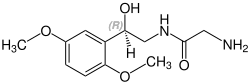 (R)-midodrine CAS number: 133163-25-4 |
 (S)-midodrine CAS number: 133267-39-7 |
Synthesis
[edit]Acylation of 1,4-dimethoxybenzene with chloroacetyl chloride gives the chloroketone 2. The halogen is then converted to the amine 3 by any set of standard schemes, and the ketone reduced to an alcohol with borohydride (4).[24] Acylation of the amino group in this last intermediate with chloroacetyl chloride affords the amide 5. The halogen is then displaced with azide and the resulting product 6 reduced catalytically to the glycinamide, midodrine (7).[25]

History
[edit]Midodrine was discovered by 1971.[6] It was approved in the United States by the Food and Drug Administration (FDA) in 1996 for the treatment of dysautonomia and orthostatic hypotension.[citation needed]
In August 2010, the FDA proposed withdrawing this approval because the manufacturer, Shire plc, failed to complete required studies after the medicine reached the market.[29][30] In September 2010, the FDA reversed its decision to remove midodrine from the market and allowed it to remain available to patients while Shire plc collected further data regarding the efficacy and safety of the drug.[31] Shire announced on September 22, 2011, that it was withdrawing completely from supplying midodrine. Midodrine remains available as a generic drug.[32]
Society and culture
[edit]Names
[edit]Midodrine is the generic name of the drug and its INN, BAN, and DCF, while its DCIT is midodrina.[6][33][34] In the case of the hydrochloride salt, its generic name is midodrine hydrochloride and this is its USAN, BANM, and JAN.[6][34] The drug is also known by its developmental code names ST-1085 and TS-701.[6][34][35] Midodrine has been sold under brand names including Amatine, Gutron, Midamine, Midon, and ProAmatine, among others.[6][34]
Research
[edit]Midodrine was under development for the treatment of chronic fatigue syndrome, but no recent development for this indication has been reported.[5]
References
[edit]- ^ a b c d e f g h i j k l m "Midodrine: Uses, Interactions, Mechanism of Action". DrugBank Online. 31 December 1992. Retrieved 1 August 2024.
- ^ "Proamatine- midodrine hydrochloride tablet". DailyMed. Retrieved 14 August 2021.
- ^ a b c d e f g h i j k l m "ProAmatine® (midodrine hydrochloride) Tablets" (PDF). fda.gov. Food and Drug Administration. Retrieved 28 August 2024.
- ^ a b c d e f g Gilden JL (2004). "Midodrine and Other Sympathomimetics". Primer on the Autonomic Nervous System. Elsevier. pp. 413–415. doi:10.1016/b978-012589762-4/50113-4. ISBN 978-0-12-589762-4.
- ^ a b "GE Healthcare". AdisInsight. 21 October 2021. Retrieved 28 August 2024.
- ^ a b c d e f Elks J (2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer US. p. 824. ISBN 978-1-4757-2085-3. Retrieved 30 August 2024.
- ^ Izcovich A, González Malla C, Manzotti M, Catalano HN, Guyatt G (September 2014). "Midodrine for orthostatic hypotension and recurrent reflex syncope: A systematic review". Neurology. 83 (13): 1170–1177. doi:10.1212/WNL.0000000000000815. PMID 25150287. S2CID 5439767.
- ^ Chen JJ, Han Y, Tang J, Portillo I, Hauser RA, Dashtipour K (December 2018). "Standing and Supine Blood Pressure Outcomes Associated With Droxidopa and Midodrine in Patients With Neurogenic Orthostatic Hypotension: A Bayesian Meta-analysis and Mixed Treatment Comparison of Randomized Trials". The Annals of Pharmacotherapy. 52 (12): 1182–1194. doi:10.1177/1060028018786954. PMID 29972032. S2CID 49674644.
- ^ Prakash S, Garg AX, Heidenheim AP, House AA (October 2004). "Midodrine appears to be safe and effective for dialysis-induced hypotension: a systematic review". Nephrology, Dialysis, Transplantation. 19 (10): 2553–2558. doi:10.1093/ndt/gfh420. PMID 15280522.
- ^ Karwa R, Woodis CB (April 2009). "Midodrine and octreotide in treatment of cirrhosis-related hemodynamic complications". The Annals of Pharmacotherapy. 43 (4): 692–699. doi:10.1345/aph.1L373. PMID 19299324. S2CID 207263346.
- ^ Narasimhan B, Aggarwal D, Satish P, Kantharia B, Aronow WS (October 2022). "Postural orthostatic tachycardia syndrome: pathophysiology, management, and experimental therapies". Expert Opin Investig Drugs. 31 (10): 1017–1025. doi:10.1080/13543784.2022.2121697. PMID 36094001.
- ^ Vasavada AM, Verma D, Sheggari V, Ghetiya S, Chirumamilla PC, Kotak RA, et al. (May 2023). "Choices and Challenges With Drug Therapy in Postural Orthostatic Tachycardia Syndrome: A Systematic Review". Cureus. 15 (5): e38887. doi:10.7759/cureus.38887. PMC 10259876. PMID 37313107.
- ^ Benarroch EE (December 2012). "Postural tachycardia syndrome: a heterogeneous and multifactorial disorder". Mayo Clin Proc. 87 (12): 1214–1225. doi:10.1016/j.mayocp.2012.08.013. PMC 3547546. PMID 23122672.
- ^ "Midodrine - FDA prescribing information, side effects and uses". Drugs.com. Retrieved 30 June 2022.
- ^ "Midodrine (Oral Route) Side Effects - Mayo Clinic". www.mayoclinic.org.
- ^ Costa-Pinto R, Jones DA, Udy AA, Warrillow SJ, Bellomo R (December 2022). "Midodrine use in critically ill patients: a narrative review". Crit Care Resusc. 24 (4): 298–308. doi:10.51893/2022.4.R. PMC 10692611. PMID 38047013.
- ^ Cruz DN (May 2000). "Midodrine: a selective alpha-adrenergic agonist for orthostatic hypotension and dialysis hypotension". Expert Opin Pharmacother. 1 (4): 835–840. doi:10.1517/14656566.1.4.835. PMID 11249519.
- ^ McClellan KJ, Wiseman LR, Wilde MI (January 1998). "Midodrine. A review of its therapeutic use in the management of orthostatic hypotension". Drugs Aging. 12 (1): 76–86. doi:10.2165/00002512-199812010-00007. PMID 9467688.
- ^ "DailyMed - MIDODRINE HCL- midodrine hydrochloride tablet". DailyMed. Retrieved 5 January 2023.
- ^ "Midodrine". PubChem. Retrieved 1 August 2024.
- ^ "Desglymidodrine". PubChem. Retrieved 1 August 2024.
- ^ "Metabolite desglymidodrine". DrugBank Online. Retrieved 1 August 2024.
- ^ Rote Liste Service GmbH (Hrsg.): Rote Liste 2017 – Arzneimittelverzeichnis für Deutschland (einschließlich EU-Zulassungen und bestimmter Medizinprodukte). Rote Liste Service GmbH, Frankfurt/Main, 2017, Aufl. 57, ISBN 978-3-946057-10-9, S. 196.
- ^ Moreau P, Finiels A, Meric P (20 March 2000). "Acetylation of dimethoxybenzenes with acetic anhydride in the presence of acidic zeolites". Journal of Molecular Catalysis A: Chemical. 154 (1): 185–192. doi:10.1016/S1381-1169(99)00373-8. ISSN 1381-1169.
- ^ Cao T, Martini ML, Park K, Kaniskan HÜ, Jin J (1 January 2022). "8.02 - Pyrimidines and Their Benzo Derivatives". In Black DS, Cossy J, Stevens CV (eds.). Comprehensive Heterocyclic Chemistry IV. Oxford: Elsevier. pp. 86–228. doi:10.1016/B978-0-12-818655-8.00041-X. ISBN 978-0-12-818656-5.
- ^ DE 2506110, Zoelss G, "Phenylethanolamine derivs prepn. - by reducing azides, useful as hypertensives", issued 21 April 1983, assigned to Lentia GmbH.
- ^ K. Wismayr et al., AT 241435; eidem, U.S. patent 3,340,298 (1965, 1967 both to Chemie Linz Ag).
- ^ Zoelss & W. Karl-Anton Ing DE 2523735 (1974 to Lentia GmbH).
- ^ U.S. proposes withdrawal of Shire hypotension drug, 16 August 2010.
- ^ O'Riordan M. "FDA recommends withdrawal of midodrine". Food and Drug Administration. FDA proposes withdrawal of low blood pressure drug [press release]. August 16, 2010. TheHeart.org. Retrieved 1 April 2011.
- ^ Midodrine (ProAmatine, generic) Proposed Market Withdrawal – Update Archived 28 March 2012 at the Wayback Machine 10 September 2010.
- ^ Shire plc. "Shire Provides Update on ProAmatine® (midodrine HCl)". www.prnewswire.com (Press release).
- ^ Morton IK, Hall JM (2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Netherlands. p. 137. ISBN 978-94-011-4439-1. Retrieved 30 August 2024.
- ^ a b c d Schweizerischer Apotheker-Verein (2004). Index Nominum: International Drug Directory. Medpharm Scientific Publishers. p. 805. ISBN 978-3-88763-101-7. Retrieved 30 August 2024.
- ^ Schlesser JL (1990). Drugs Available Abroad: A Guide to Therapeutic Drugs Available and Approved for Use Outside the U. S. Gale Research. pp. 139, 356. ISBN 978-0-8103-7177-4. Retrieved 30 August 2024.
External links
[edit]![]() Media related to Midodrine at Wikimedia Commons
Media related to Midodrine at Wikimedia Commons