
| |
 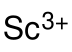
| |
| Names | |
|---|---|
| Other names
Scandium(III) acetate
Scandium ethanoate Scandium(III) ethanoate | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.021.159 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| Properties | |
| C6H9O6Sc | |
| Molar mass | 222.088 g·mol−1 |
| Appearance | white solid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Scandium acetate is an compound, with the chemical formula of Sc(CH3COO)3. It exists in the anhydrous and the hydrate forms. It can be obtained by reacting scandium hydroxide or scandium oxide with acetic acid.[1][2] It is a colorless, water-soluble solid. It decomposes into scandium oxide at high temperature. It can be used to prepare other scandium-containing materials.[3]
The structure of the anhydrous form was determined by X-ray crystallography. It consists of a chain of octahedral Sc(III) centers linked by bridging acetate ligands.[1]
References
[edit]- ^ a b Fuchs, Robert; Strähle, Joachim (1984). "Die Kristallstruktur von Sc(CH3COO)3, ein Metall(III)acetat mit Kettenstruktur" [Crystal Structure of Sc(CH3COO)3, a Metal(III) Acetate with a Chain Structure]. Zeitschrift für Naturforschung B. 39 (12): 1662–1663. doi:10.1515/znb-1984-1206.
- ^ Krasil'Nikov, V. N.; Gyrdasova, O. I.; Baklanova, I. V.; Perelyaeva, L. A.; Zhilina, E. F.; Vovkotrub, É. G. (2012). "Structure and luminescence properties of nanostructured solid-state solutions of Sc1−xEux(CH3CO2)3". Theoretical and Experimental Chemistry. 48 (2): 113–117. doi:10.1007/s11237-012-9247-9.
- ^ Xin, Chengrong; Zhang, Qilong; Yang, Hui (2013). "Synthesis and Properties Study of BaTi0.95Sc0.05O3−δ Nanopowders and Ceramics". Piezoelectrics & Acoustooptics (in Chinese). 35 (3): 412–415.